Chapter contents:
1.Brachiopoda
–– 1.1 Brachiopod Classification
–– 1.2 Brachiopods vs. Bivalves
–– 1.3 Brachiopod Paleoecology ←
–– 1.4 Brachiopod Preservation
Above Image: Animal forms; a second book of zoology (1902), Figure 43: Animals of Uncertain Relationships. Source: Wikimedia Commons (Public Domain).
Overview
Brachiopods are solitary creatures that inhabit the seafloor across a variety of habitats. Because they are sessile (unmoving), they filter food particles and nutrients out of the water. Like many marine invertebrates, brachiopods have an embryonic, larval, and juvenile stage. Larvae float in the water column before they settle and attach to substrate on the ocean floor.
Brachiopods face several challenges living on the seafloor and each habitat poses its own challenges. Lacking the ability to move around, some have evolved special structures to prevent being flipped over or swept away by currents and storms.
One of the main distinguishing characteristics of Phylum Brachiopoda is the muscular pedicle that they use for attachment, though not all brachiopods have them as adults (some species only have a pedicle in their juvenile form). The pedicles of different species may vary depending on the habitats that they occupy. For example, if a brachiopod species lives in rocky or reef environments, they might have a thicker and more muscular pedicle to help them attach to hard surfaces. In soft sandy or muddy environments, having a pedicle with a root-like structure might be more beneficial. Some brachiopods used their pedicle as a precautionary anchor to prevent them from being swept away in strong currents or storms.
Brachiopods lacking a pedicle altogether weighed down their shells with extra calcite to ‘recline’, either cementing themselves to hard substrates like modern oysters, or evolving spines to help keep them in place on the soft ocean floor. The long wings of spirifer brachiopods might have been an evolutionary response to also help address this problem.
Natural Predators?
Modern brachiopods have very little living tissue and thick shells, and this was almost certainly true in extinct species as well. They have therefore been considered to not be worthwhile prey, given the small reward. However, recent observations on modern brachiopod populations reveal they may not be completely safe from predation. Repair scars, which indicate survival from an attack by a predator, highlight moderate levels of predation by migrating birds, echinoderms, crustaceans, fish, and snails. Although brachiopods may not be a preferred dinner choice, they are occasionally eaten and aren’t completely safe from predation.
To assist with defence against predators, ancient brachiopods built up defenses such as spines at the same time when durophagous (shell-crushing) predators diversified during the Paleozoic. Spines not only serve as direct protection, but they also provide the illusion that shells seem larger than they are, which may prevent a predator from attacking. Some brachiopods also developed thicker shells and ornamentation, such as coarse ribs, to deter potential predators.
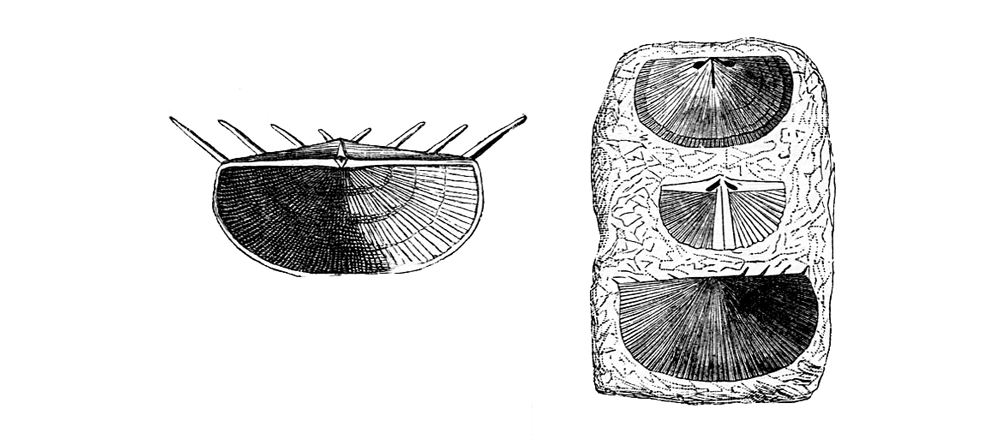
Hinge spine diversity. Left: Chonetes striatellus from the Silurian of Sweden. Right: Chonetes sarcinulatus from the Devonian of Germany. (Zittel 1913, Fig. 578) (Public Domain)
Fossil specimen of a productid brachiopod replaced by quartz from the Permian Glass Mountains of southwest Texas (PRI 76879). From the collections of the Paleontological Research Institution. The longest dimension is 4 cm in length.
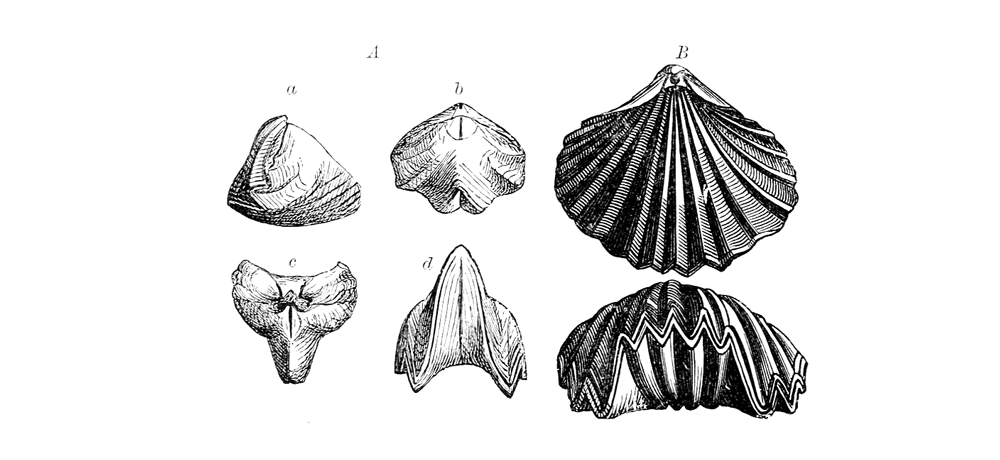
Brachiopods with coarse ribbing. A: Rhynconella loxia of the Upper Jurassic of Russia. B: Cylothyris quadriplicata of the Middle Jurassic of Germany (Zittel, 1913, Fig. 592). (Public Domain)
Encrusting Communities
Although predatory traces are rare on brachiopod fossils, their shells were commonly a substrate for encrusting organisms like corals and bryozoans. In some cases, the spines of brachiopods seemed to attract bryozoans, forming a camouflage cover that allowed them to hide in plain sight.
Fossil specimen of the brachiopod Rafinesquina deltoidea from the Ordovician Trenton Group of Lewis County, New York (PRI 70590). The surface of this specimen is covered by an encrusting tabulate coral (perhaps Protarea). Specimen is from the research collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 5.5 cm.
Fossil specimen of the brachiopod Spinocyrtia granulosa with encrusting Aulopora sp. corals from the Devonian Ludlowville Formation of Erie County, New York (PRI 44051). Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York.
Fossil specimen of the brachiopod Athyris spriferoides, which is partially encrusted by bryozoans; from the Devonian Ludlowville Formation of Erie County, New York (PRI 44050). Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York.
Fossil brachiopod specimen of Ladogia meyendorfi (PRI 76920) from the Lower Devonian with encrusters on the shell surface. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Longest dimension of specimen is approximately 3.5 cm. Model by Jaleigh Pier.
Brachiopod Communities
Brachiopods were the most abundant shelly fauna on the ocean bottom during the Paleozoic. Some species "bloomed" in gregarious numbers, becoming the dominant taxa in some locations.
Small slab limestone covered by the brachiopod Cincinnetina meeki. Specimen is from the Ordovician Richmond Group of Cincinnati, Ohio and is housed in the research collections of the Paleontological Research Institution, Ithaca, New York. Length of rock is approximately 12 cm.
Specimens of the inarticulate brachiopod Obolus matinalis from the Cambrian of Polk County, Wisconsin (PRI 76740). Specimens are from the collections of the Paleontological Research Institution, Ithaca, New York. Longest dimension of specimen is approximately 7 cm.
A small slab from Oxford, Ohio covered with numerous specimens of the Ordovician brachiopod Hiscobeccus capax (PRI 70758). This specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Specimen is approximately 27 cm in length.
Community Comparison
Other preserved brachiopod communities are much more diverse. Diversity is often studied by counting the abundances and variety of species present in the same location. The sediments and other materials preserved in the rocks can indicate what the environment was like. For example, fine shales are often indicative of deep water conditions, while courser sandstones often indicate shallow water. Can you tell any differences between the Ordovician and Devonian brachiopod communities shown in the models below?
Ordovician Brachiopod Community:
Slab of fossil brachiopods from the Upper Ordovician Waynesville Formation of Warren County, Ohio (PRI 76881). Specimen from the Paleontological Research Institution, Ithaca, New York. Longest dimension is approximately 28 cm. Model by Jaleigh Pier.
During the Late Ordovician (~450 million years ago), the fossils preserved in this slab were very much alive in what is part of Ohio today. The rock is made of siltstone, indicating that the environment was likely in a deeper offshore setting, where fine sediments could have settled. The brachiopod shells are preserved in great condition, allowing rhynconellids, strophomenids, an orthid, and a spirifer to be recognized. The strophomenids are larger with several distinct species, but the spirifer is smaller and less winged than those on the Devonian slab shown below. There are also a few bryozoan species, including encrusting and branching forms, as well as several large rugose corals. Overall, this Ordovician slab demonstrates a higher diversity than the Devonian slab, although it also has much more shell hash preserved as well.
Devonian Brachiopod Community:
Rock slab of fossil brachiopods from the Upper Devonian Chemung Formation of Steuben County, New York (PRI 76878). Specimen from the Paleontological Research Collection, Ithaca, New York. Longest dimension is approximately 28 cm. Model by Jaleigh Pier.
The Late Devonian slab shown above preserved a community that existed along the New York/Pennsylvania border ~373 million years ago. Here we can identify atrypid, spirifer, strophomenid, and orthid brachiopod species. The preservation is different relative to the Ordovician slab: the shell material has long since dissolved away so we are left with molds and casts where the shells once were. A few rugose corals and possibly a crinoid or two co-existed with these brachiopods, although they were much smaller in size compared to those on the Ordovician slab. The rock is a fine-grained sandstone and may have formed slightly closer to shore than the Ordovician specimen (as indicated by the coarser sediments). There is also much more sediment relative to fossils, which tells us that either sediment accumulated much faster here, or there were just fewer living things in this environment.
These two rocks slabs reveal snapshots in time of two completely different communities. Based on the rock type, fossil preservation, and diversity present we can infer what those environments were like and how they are different. Based on your own observations of these rock slabs, can you make any additional conclusions?
References and further reading
Alexander, R.R. 2001. Functional Morphology and Biomechanics of Articulate Brachiopod Shells, in Carlson, S.J. and Sandy, M.R. ed., Brachiopods Ancient and Modern. The Paleontological Society Papers, Volume 7:145-169.
Boardman, R.S., Cheetham, A.H., and Rowell, A.J. 1987. Fossil Invertebrates. Blackwell Scientific Publications. 713 pp.
David, J.S. and Heath H. 1902. Animal Forms, a second book of zoology. New York D. Appleton and Company.
Feifarek, B.P., 1987. Spines and epibionts as antipredator defenses in the thorny oyster Spondylus americanus Hermann. Journal of Experimental Marine Biology and Ecology 105, 39-56.
Kowalewski, M., Dulai, A., Fürsich, F.T., 1998. A fossil record full of holes: the Phanerozoic history of drilling predation. Geology 26, 1091–1094.
Johnsen, S.A.L., Ahmed, M., and Leighton, L.R., 2013. The effect of spines of a Devonian productide brachiopod on durophagous predation. Palaeogeography, Palaeoclimatology, Palaeoecology 375, 30-37.
Leighton, L.R. 2001. New Directions in the Paleoecology of Paleozoic Brachiopods, in Carlson, S.J. and Sandy, M.R. ed., Brachiopods Ancient and Modern. The Paleontological Society Papers, Volume 7:185-205.
Peck, L.S. 2001. Ecology of Articulated Brachiopods, in Carlson, S.J. and Sandy, M.R. ed., Brachiopods Ancient and Modern. The Paleontological Society Papers, Volume 7:171-183.
Peck, L.S. 2001. Physiology, in Carlson, S.J. and Sandy, M.R. ed., Brachiopods Ancient and Modern. The Paleontological Society Papers, Volume 7:89-104.
A. Selden ed., Treatise on Invertebrate Paleontology, Part H, Brachiopoda Revised, Volume 6. The University of Kansas and Geological Society of America. 3226 pp.
Signor, P.W., Brett, C.E., 1984. The mid-Paleozoic precursor to the Mesozoic marine revolution. Paleobiology 10, 229–245.
2018. How is biodiversity produced? Examining speciation processes during the GOBE. Lethaia, Volume 51: 165– 172.
Stigall, A.L., Edwards, C.T., Freeman, R.L., Rasmussen, C.M. 2019. Coordinated biotic and abiotic change during the Great Ordovician Biodiversification Event: Darriwilian assembly of early Paleozoic building blocks. Palaeogeography, Palaeoclimatology, Palaeoecology,
Volume 530: 249-270
Tyler, C.L., Leighton, L.R., Carlson, S.J., Huntley, J.W., and Kowalewski, M., 2013. Predation on modern and fossil brachiopods: Assessing chemical defenses and palatability. Palaios 28, 724-735.
Williams, A. and Carlson, S. J. 2007. Affinities of Brachiopods and Trends in their Evolution, in P. A. Selden ed., Treatise on Invertebrate Paleontology, Part H, Brachiopoda Revised, Volume 6. The University of Kansas and Geological Society of America. 3226 pp.
Willman, S., 2007. Testing the role of spines as predatory defense. Journal of Shellfish Research 26, 261–266.
Zittel, K.A. 1913. Brachiopoda, in Eastman, C.R. ed., Text-Book of Paleontology. Macmillan and Co. Limited, London. 839 pp.
Usage

Unless otherwise indicated, the written and visual content on this page is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. This page was written by Jaleigh Q. Pier. See captions of individual images for attributions. See original source material for licenses associated with video and/or 3D model content.



