Chapter contents:
Class Bivalvia: Introduction and Morphology
– 1. Bivalve Phylogeny and Classification
– 2. Bivalve Ecology and Paleoecology ←
– 3. Evolutionary History of Bivalves
– 4. References and Further Reading
Image above: Mussels attached to rocks are exposed during low tide along the central California coast. Photograph by Jonathan R. Hendricks.
Life Habits
Bivalves live in a wide variety of habitats and differ widely in their ecological relationships. Although most bivalves are marine, a large number of living species inhabit freshwater. Although most marine and freshwater species are suspension feeders – filtering food particles from water pumped over their gills – bivalves also make a living in many other ways, including as predators, by eating wood, or keeping photo- or chemosymbiotic microorganisms in their tissues. Most bivalves live on or in bottom sediments, including boring in wood or rock, but they also cement or byssally attach to rocks, seafans, and other bivalves, nestle in rock crevices, or live commensally on the bodies of (or in the burrows of) other invertebrates.
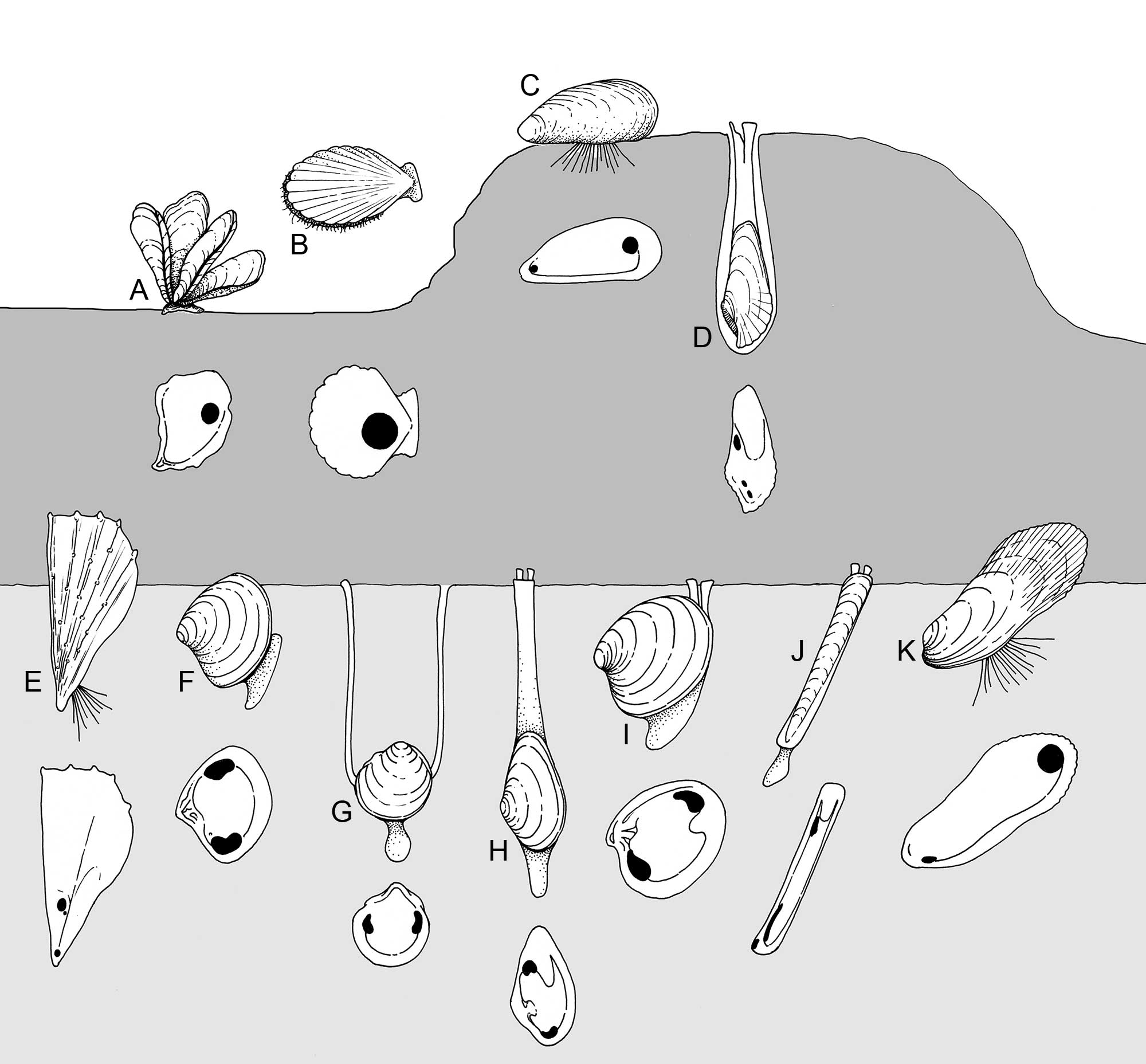
Diverse lifemodes of various benthic bivalves, with the interior of one valve showing position of adductor muscles and pallial line. (Light gray = soft substrate; dark gray = hard substrate) A. Crassostrea (Ostreidae), cemented. B. Argopecten (Pectenidae), epibenthic swimmer. C. Mytilus (Mytilidae), epibyssate. D. Pholas (Pholadidae), rock-borer. E. Pinna (Pinnidae), shallow burrower. F. Astarte (Astartidae), shallow burrower. G. Phacoides (Lucinidae), deep burrower with mucus siphons H. Mya (Myidae), deep burrower with fused mantle siphons. I. Mercenaria (Veneridae), shallow burrower. J. Ensis (Pharidae), deep burrower with fused mantle siphons. K. Modiolus (Mytilidae), endobyssate. (Drawing by Christi Sobel; based on Stanley (1970), and other sources.)

Lifestyles of different types of bivalves. Image by Franz Anthony and Dr. Katie Collins. Copyright Franz Anthony; used with permission.
Enigmonia, the mangrove jingle shell, lives on the underside of mangrove leaves or high on seawalls in the Indo-Pacific region where it is bathed only in ocean spray; it is the closest that we have to a “terrestrial bivalve.”

The terrestrial mangrove jingle shell clam Enigmonia aenigmatica (Family Anomiidae) at Lim Chu Kang, Singapore. Photograph by Ria Tan (flickr; Creative Commons Attribution-NonCommercial-noDerivs 2.0 Generic license).

Modern specimens of the mangrove jingle shell Enigmonia aenigmatica from the Philippines (PRI 90588). Specimens are from the collections of the Paleontological Research Institution, Ithaca, NY. Photograph by Vicky Wang.
Reproduction and Life History
Bivalve reproduction and life history are also diverse. Most species have larvae that float for days to months in the plankton, feeding or not. Others brood their larvae in their gills and lack a planktonic stage entirely. The larval shell (the prodissoconch) of modern and fossil species reflects these life history patterns, with larger shells of fewer spirals characterizing brooders or non-plankton-feeding (nonplanktotrophic or direct-developing) species, and smaller shells with more spirals characterizing planktotrophic species.
"Bivalve Veliger" by George von Dassow (YouTube).
Paleoecology
Because bivalves usually live in close association with their substrates, their shells frequently closely reflect their mode of life (see image above). This means that shell shape of fossil bivalves can be used to infer their paleoecology and mode of life. For example, the presence of a pallial sinus on the interior surface of a bivalve indicates that the organism had siphons, thus likely burrowed in soft substrate (see example below of the deep pallial sinus on the specimen of Mya truncata).
An irregularly-shaped cementation scar on one valve as in oysters is a clear indication of epibenthic attachment. For example, see how the oyster below is cemented to the fossil coral.
Fossil specimen of the scleractinian coral Scolymia lacera associated with an oyster that grew upon the coral (PRI 76871). Specimen is from the Plio-Pleistocene of Sarasota County, Florida and is part of the research collection of the Paleontological Research Institution, Ithaca, New York. Length of specimen is approximately 11 cm (Sketchfab; Creative Commons 0 license).
A gap in the ventral or anterior margin of an otherwise tightly closing bivalve suggests a byssus, and thus likewise attachment to hard substrate above the bottom.

Byssal notch (or, byssal gap) in a specimen of Arca wagneriana from the upper Pliocene Tamiami Fm. (Pinecrest Beds) of Sarasota County, Florida (collections of the Paleontological Research Institution, Ithaca, NY). Photograph by Warren D. Allmon.
Strong shell sculpture could indicate either burrowing (often used to stabilize a bivalve in shifting sand or rasping into wood or soft rock) or attachment (for example, long spines in Spondylus or Arcinella (below) serve as substrate for algae, sponges, etc. for camouflage and also for defense from predators).
Arcinella cornuta from the Plio-Pleistocene of Florida (PRI 44151). Specimen is from the research collection of the Paleontological Research Institution, Ithaca, NY.
Several different groups of bivalves have formed reefs or reef-like masses at different times in geological history. During the Cretaceous, rudists were important builders of reef-like mounds in the super-warm waters of the equatorial Tethys Ocean and the Western Interior Seaway of North America.

Portion of a rudist bivalve reef from the Early Cretaceous Edwards Formation of Bosque County, Texas. Rudist species present in the reef include Eoradiolites robustus and Eoradiolites davidsoni. Specimen is on display at the Denver Museum of Nature and Science (DMNH 8025, 8026). Photograph by Jonathan R. Hendricks.
During the Neogene, ostreid oysters became important constructors of rigid reef structures, and they continue to do so in the present.

A conservation researcher sampling a modern oyster reef. Photograph by Jansen A. Smith.
Bivalves are host to many other invertebrates. Pen shells (Pinna) and pearl oysters (Pinctada) often have commensal, male/female pairs of “pea crabs” (Pinnotheridae) living within their spacious mantle cavities. Giant Clams (Tridacna) and Spiny Oysters (Spondylus) often have burrowing sponges and bivalves (especially Gastrochaenidae) or nestling bivalves in holes and crevices of their thick shells. Tridacna harbors photosynthetic algal cells in its mantle tissues, which provide a supplemental energy source to the clam.

And like any other hard substrate in water, the shells of bivalves, especially non-burrowing species, are almost always covered by fouling communities of algae, sponge, barnacles, limpets, and other attached organisms (including other bivalves).




