Chapter by:
Warren D. Allmon (Paleontological Research Institution, Ithaca, New York) and Paula M. Mikkelsen (Integrative Research Center, Field Museum of Natural History, Chicago, Illinois).
This chapter was first publicly shared on October 30, 2020; it was last updated on February 14, 2022.
Chapter citation:
Allmon, W. D., and P. M. Mikkelsen. 2020. Bivalvia. In: The Digital Encyclopedia of Ancient Life. https://www.digitalatlasofancientlife.org/learn/mollusca/bivalvia/
Image above: Examples of different types of modern and fossil bivalves. Photographs by Jonathan R. Hendricks.
Acknowledgments:
For assistance with illustrations, we are grateful to Alexandra Allmon, Franz Anthony, James Crampton, Katy Estes-Smargiassi, Liz Harper, Jon Hendricks, Carole Hickman, Linda Ivany, Claudia Johnson, Carlie Pietsch, Elizabeth Petsios, Gary Rosenberg, Leslie Skibinski, Christi Sobel, Chelsea Steffes, Jennifer Tegan, Vicky Wang, and Alex Zimmerman. For comments on the text we thank Chris McRoberts.
Chapter contents:
Class Bivalvia: Introduction and Morphology ←
– 1. Bivalve Phylogeny and Classification
– 2. Bivalve Ecology and Paleoecology
– 3. Evolutionary History of Bivalves
– 4. References and Further Reading
Virtual Collection:
A virtual collection of interactive 3D models of bivalve specimens is associated with this chapter. Access here.
Snapshot: Bivalvia
Phylum Mollusca, Class Bivalvia
Common names of representatives: clams, scallops, oysters, mussels.
Habitat(s): marine (salt water), freshwater (lakes, rivers, and streams).
Feeding type(s): mostly suspension feeders; some deposit feeders and carnivores
Geological range: Cambrian to today.
Clade defining feature(s): two hinged shells surrounding a body with large stomach, foot, and filamentous gills; no head or radula.
Overview
Bivalves (also known as “pelecypods” or, in older literature, “lamellibranchs”) are a very diverse and abundant group of mollusks. They live today throughout the world’s oceans and fresh waters, where they are of major ecological importance as a food source for other organisms and for their water-filtering capabilities. They have a long and rich fossil record that illustrates their complex evolutionary history. Bivalves are also of major economic importance to humans, as sources of food and other products, and as damaging invasive species.
Bivalves are members of the phylum Mollusca, which also includes cephalopods (squids, octopuses, nautiloids, and ammonoids), gastropods (snails, slugs, and nudibranchs), scaphopods (tusk shells), polyplacophorans (chitons), the extinct rostroconchs, and three “minor” groups, the monoplacophorans, Caudofoveata, and Solenogastres (the latter two are generally collectively called “aplacophorans”). At first glance, bivalves do not appear to have much in common with these other groups besides having a shell made of calcium carbonate, which most possess. In fact, the only major feature that all mollusks possess is a sheet of tissue covering the body called the mantle, which (in most) covers the viscera and gills, and secretes the shell. Unlike all other mollusks, bivalves lack a distinct head with all of its associated organs (tentacles, radula, etc.), and have a pair of shells (called valves), which enclose a laterally compressed soft body, and are usually connected by a hinge and a flexible ligament. All bivalves live in or very close to the water, and have lamellar gills for respiration (which are also used for feeding in many species).
Bivalves include clams, scallops, mussels, oysters, and their relatives. There may be as many as 50,000 described living species of bivalves (estimates of the diversity of living species range from 7500 to about 50,000; see Huber, 2010; Ponder et al., 2020) and over 42,000 described fossil species (Pojeta, 1987). The oldest known fossil bivalves come from rocks of Early Cambrian age. For most of their history, and especially for the past 250 million years, bivalves have been among the most taxonomically diverse and ecologically important groups of animals in the oceans.
Bivalve Anatomy
The valve structure and soft anatomy of bivalves varies among species. The modern hard-shelled clam (also known as the quahog or cherrystone), Mercenaria mercenaria (family Veneridae), is used here as an example of a typical bivalve.
Exterior Features of the Shell
Interactive 3D model of a fossil specimen of the bivalve Mercenaria mercenaria from the Quaternary of St. Mary’s County, Maryland (PRI 76728), with external shell features annotated. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 10 cm.
Valves
As their name implies, bivalves have two shells that are called valves. The valves are made of calcium carbonate (CaCO3) in the form of the minerals aragonite and/or calcite. In most species the valves are approximately the same size, but in some they are unequal, a condition called inequivalve. The meeting of the two valves is sometimes called the commissure or shell margin. The knob-like, sometimes-pointed (and earliest-formed) part of a valve is the umbo (or “beak”, pl. umbones).
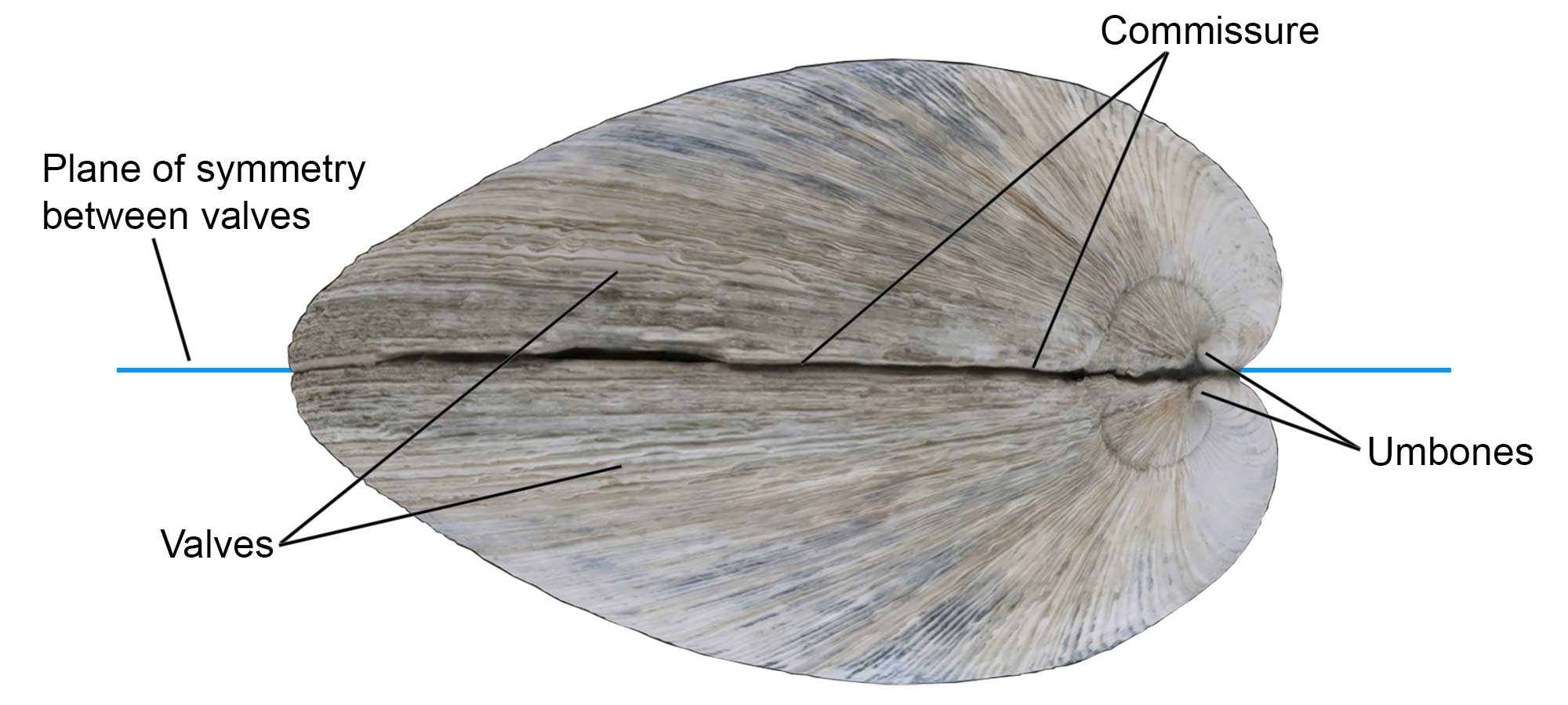
Some of the external shell features of the bivalve Mercenaria mercenaria, including the valves, commissure, and umbones. Specimen is from the Quaternary of St. Mary’s County, Maryland (PRI 76728), with external shell features annotated. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 10 cm.
In most bivalves, the plane of symmetry is between the two valves (that is, the shells lie laterally on the body, and the right and left sides are nearly symmetrical, whereas in most brachiopods it is across the valves (i.e., the valves lie dorsally and ventrally on the body).
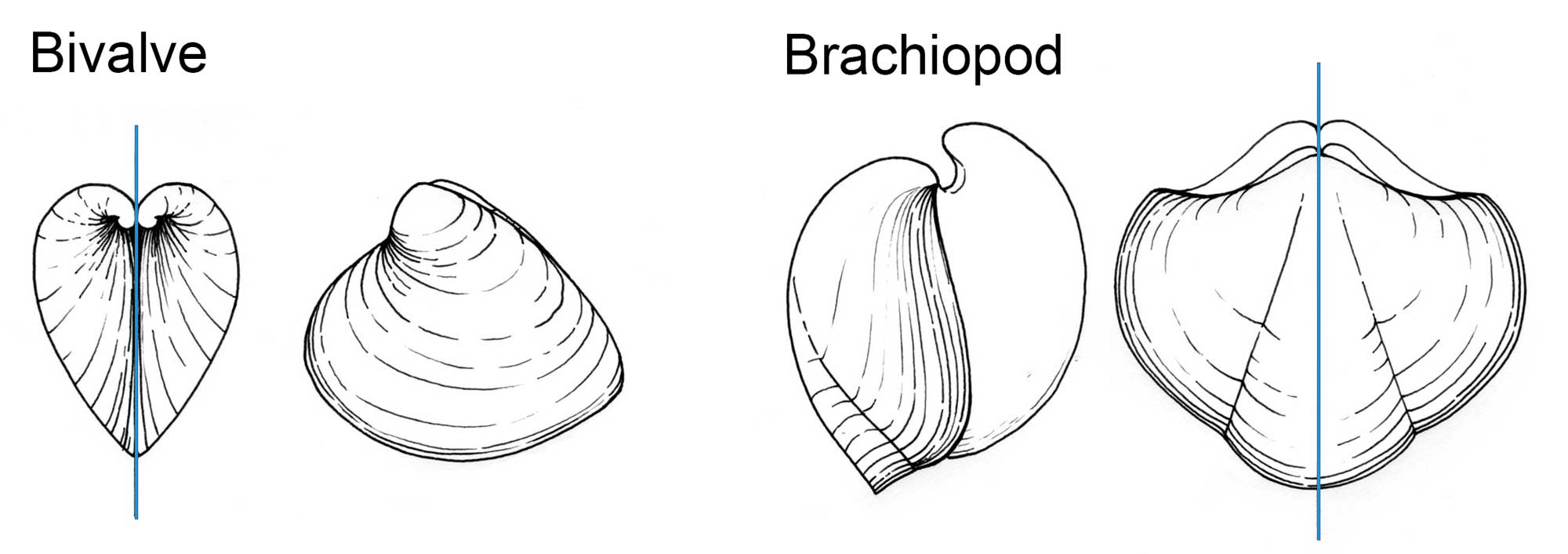
Difference between a bivalve mollusk (left two diagrams), in which the plane of symmetry (blue line) is usually between the valves and corresponds with the commissure, and a brachiopod (right two diagrams) in which the plane of symmetry is usually across the valves. Drawing by Christi Sobel.
Growth Lines
Growth lines parallel to the shell margins mark increments of growth of the shell, similar to tree rings. More elaborate sculpture common in bivalves include commarginal (parallel to the shell edge, like growth lines), radial (from umbo to ventral edge), or in a few, oblique elements.
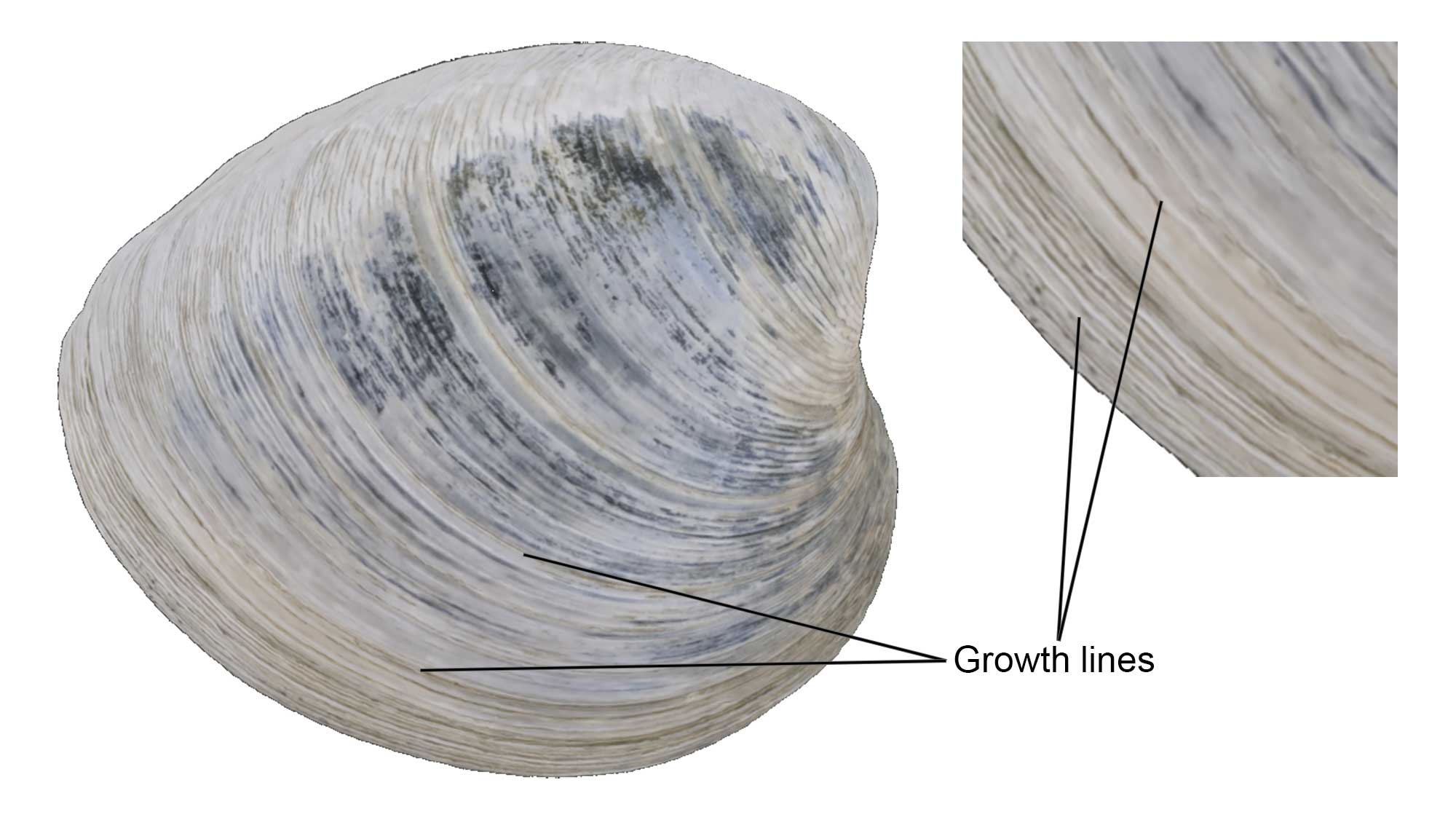
Growth lines on a specimen of Mercenaria mercenaria from the Quaternary of St. Mary’s County, Maryland (PRI 76728), with external shell features annotated. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 10 cm.
Lunule and Escutcheon
The lunule and escutcheon are impressed external features anterior or posterior, respectively, to the umbo.
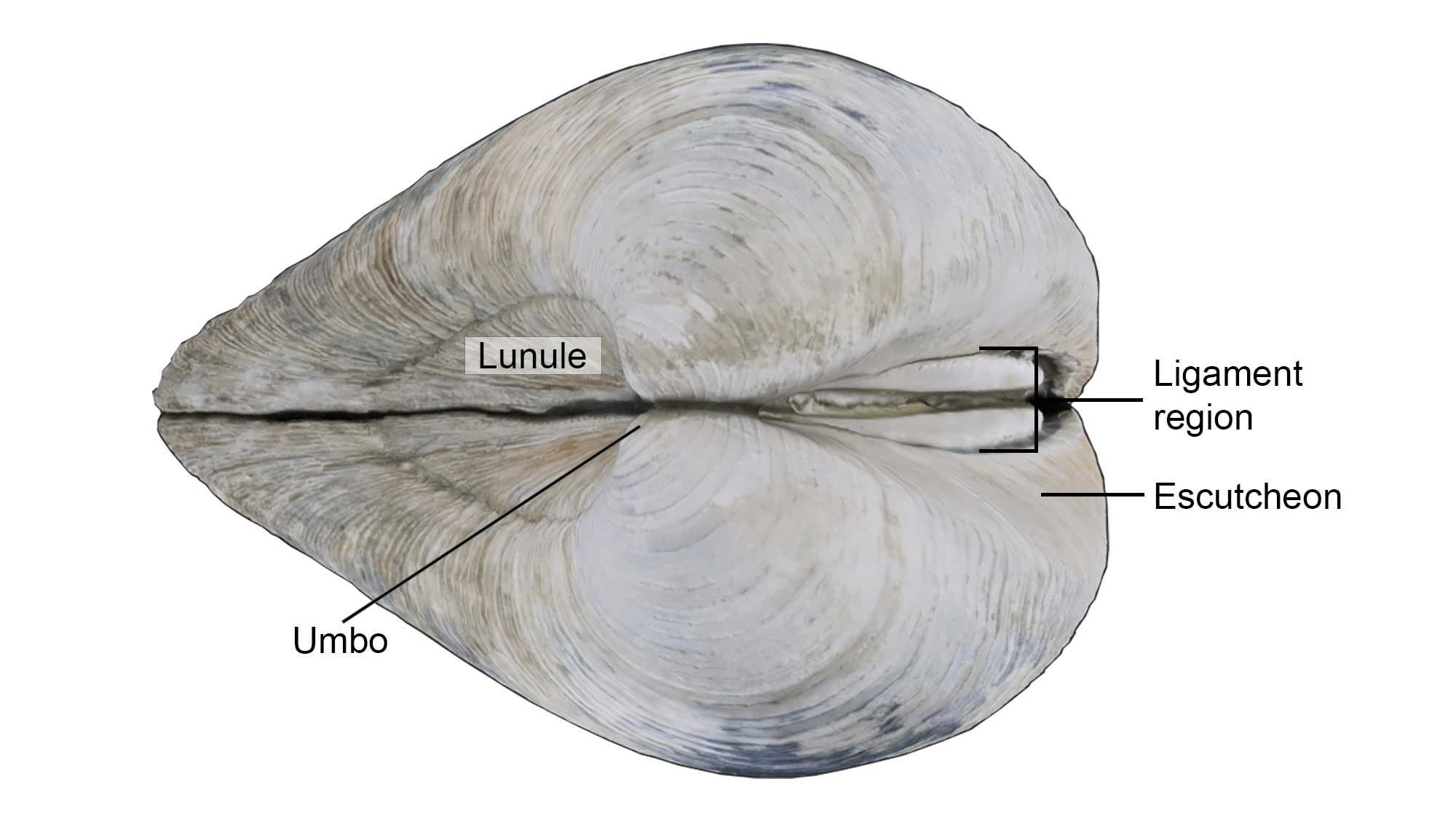
Some of the external shell features of the bivalve Mercenaria mercenaria, including the lunule, escutcheon, and umbo. Specimen is from the Quaternary of St. Mary’s County, Maryland (PRI 76728), with external shell features annotated. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 10 cm.
Larval Shell (Prodissoconch)
The larval shell, which is frequently retained on the umbo in older individuals, is called the prodissoconch.

Scanning electron photomicrograph of the prodissoconch of the modern bivalve Nuculana acuta. Image provided by Paul Harnik.
Bivalve shell shapes
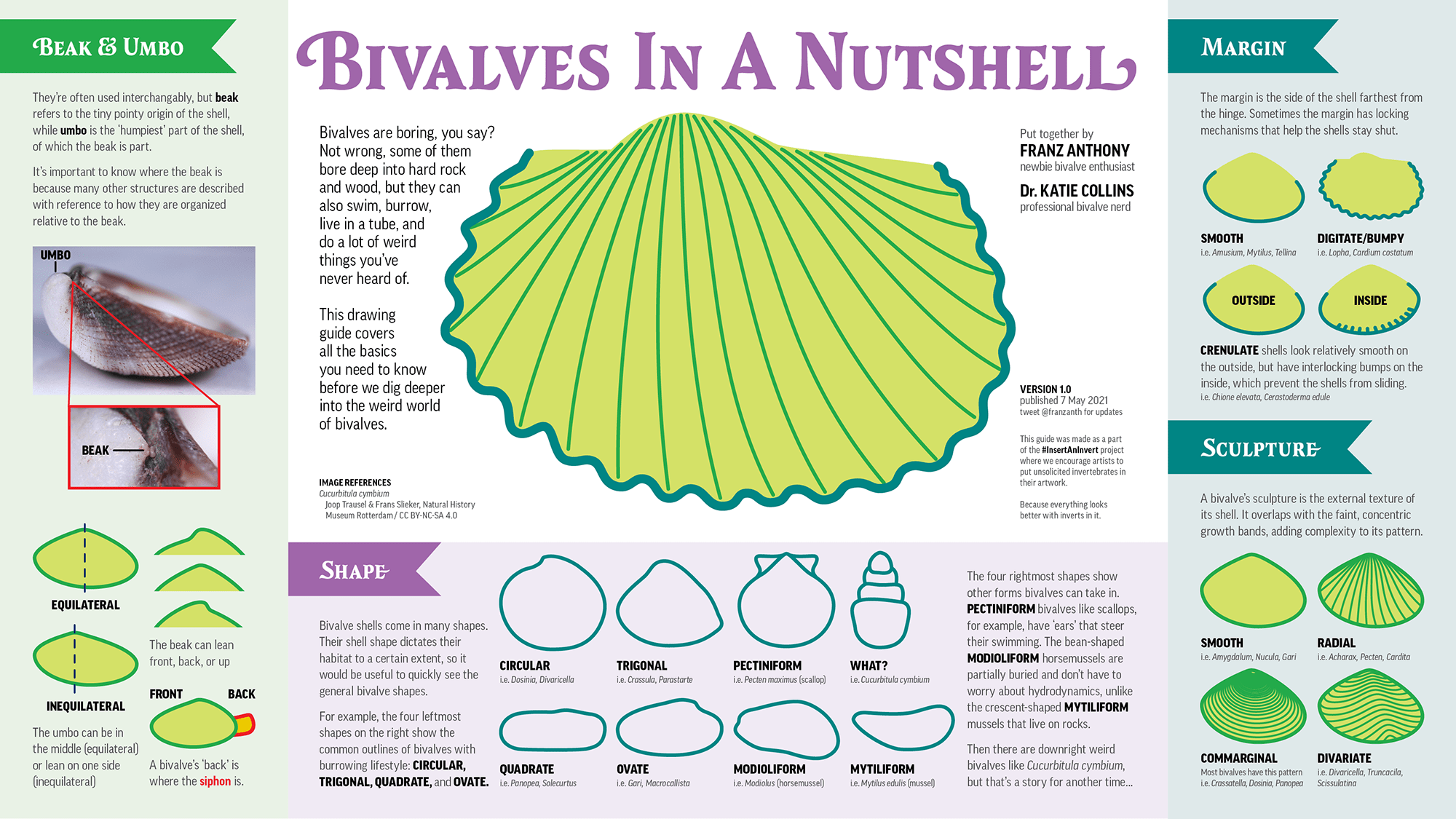
Varied aspects of the shapes of bivalve shells. Image by Franz Anthony and Dr. Katie Collins. Copyright Franz Anthony; used with permission.
Interior Features of the Shell
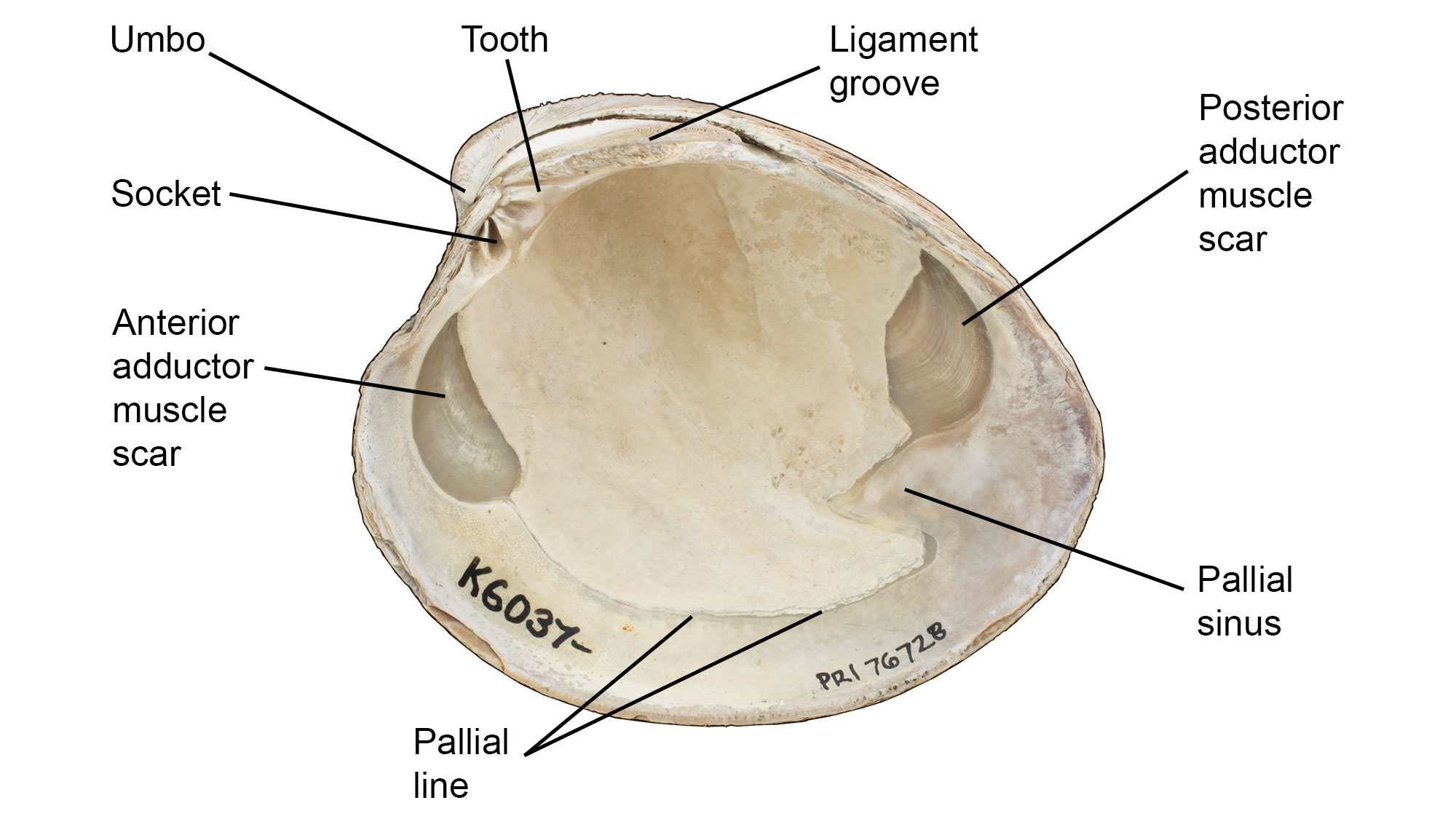
Interior features of a valve of Mercenaria mercenaria. Specimen is from the Quaternary of St. Mary’s County, Maryland (PRI 76728), with external shell features annotated. Specimen is from the collections of the Paleontological Research Institution, Ithaca, New York. Width of specimen is approximately 10 cm.
Muscle Scars
Bivalves use muscles to close their shells. The areas where the muscles connected with the inner surfaces of the shell are represented by muscle scars. These scars indicate the position of the muscles in the bivalve's body, as well as the overall sizes of the muscles (see additional discussion of the muscles themselves below).
Pallial Line
The pallial line represents the attachment of the soft tissues to the interior of the shell; an embayment (pallial sinus) is present posteriorly in siphon-bearing bivalves, into which the siphons contract when the shell is closed.
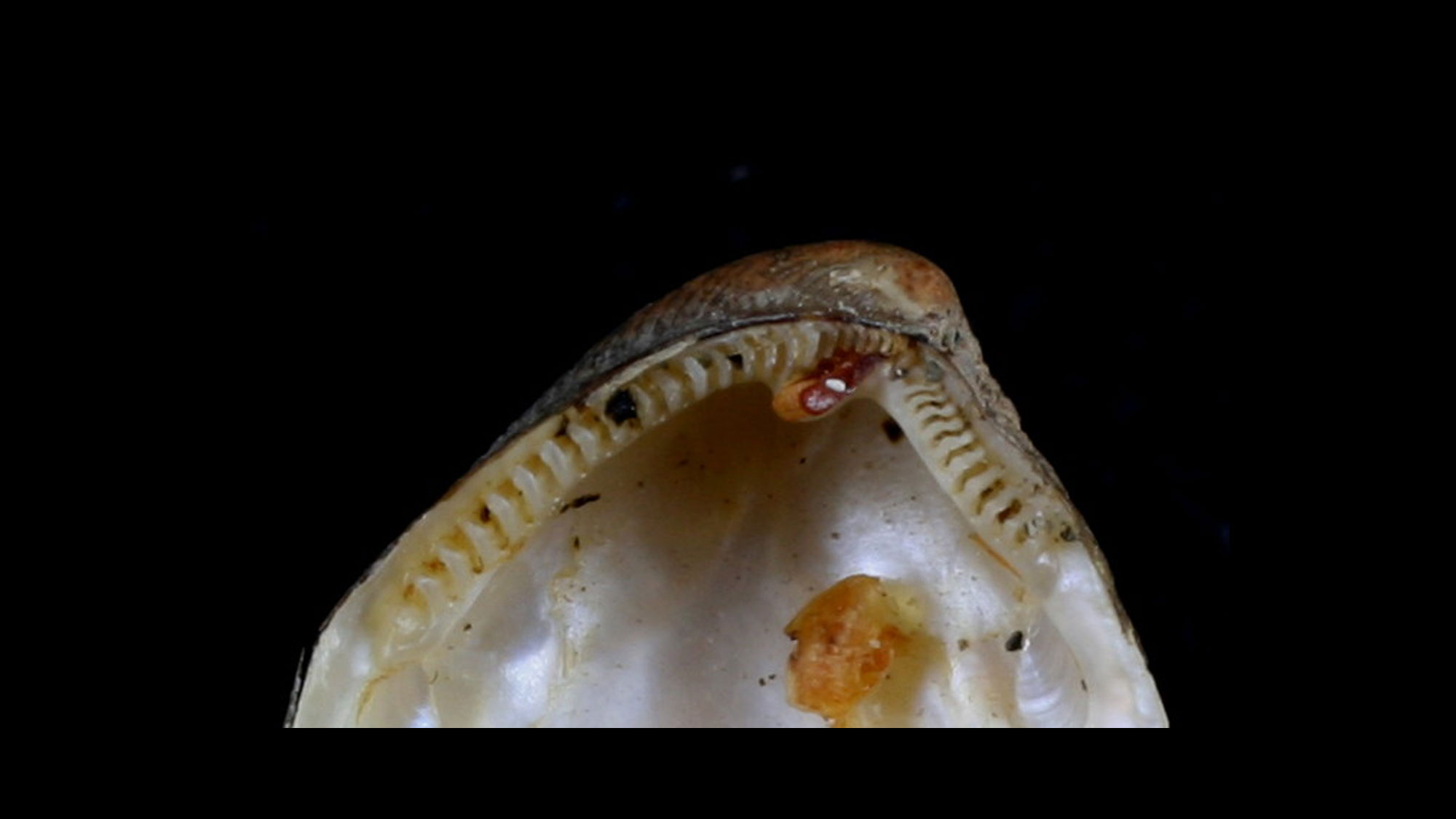
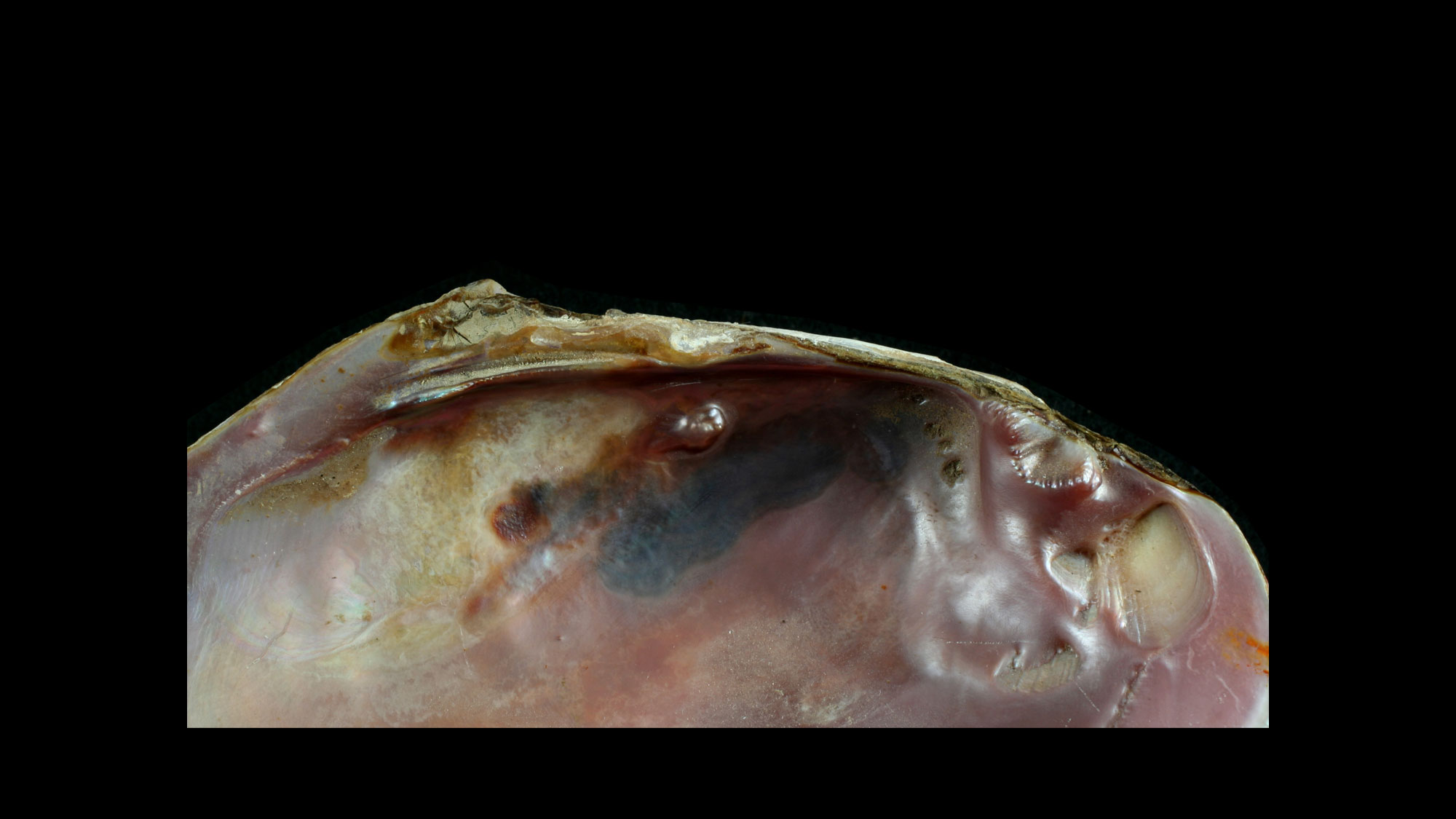
Hinge
A bivalve's two valves are connected by a hinge with a series of interlocking teeth and sockets that keep the valves aligned, especially when opening and closing. Various arrangements of hinge teeth have been named (see images below). Small denticles or crenulations along the lateral and ventral shell margins also serve to keep the valves aligned upon closure.














Ligament
An elastic ligament connects the two valves, allowing them to spring open when the adductor muscles relax.

Hinge ligament (dried) of a mussel shell. Photograph by "Shellnut" (Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
Orientation of the Shell
In Mercenaria (and most species), the umbo points toward the anterior and the ligament toward the posterior (this is not necessarily the case with other bivalves). If you hold a Mercenaria with both valves with the umbo pointing away from you and the shell margins down, the valve on the right is conventionally referred to as the right valve, and the valve on the left is the left valve.
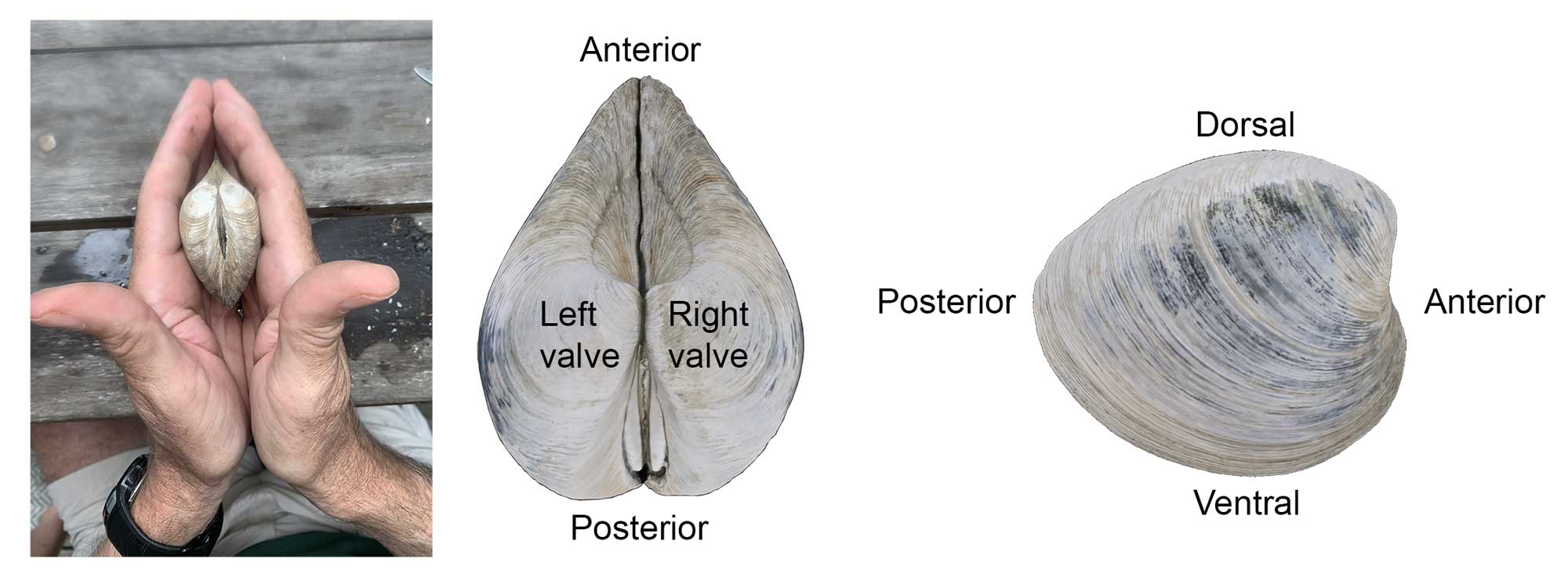
The left and right valves of a Mercenaria shell may be determined by holding the shell with the umbo pointing away from you and the ventral shell margin (i.e., most recent area of shell growth) pointing downward. The valve on the left is the left valve and the valve on the right is the right valve. In most bivalves (including Mercenaria), the umbo points towards the anterior side and the ligament and escutcheon towards the posterior side. The side of the shell with the hinge is dorsal and the opposite side (i.e., margin of shell growth) is ventral.
Shell Microstructure
Growth occurs only at the margin of the shell, where new shell material is added by folds at the edge of the mantle (see below) (although thickening of the shell occurs on its inner surface across the upper surface of the mantle). Growth increments form the growth lines that are usually visible on the outside of the shell, and also when the shell is sliced across those lines, from umbo to the shell margin.
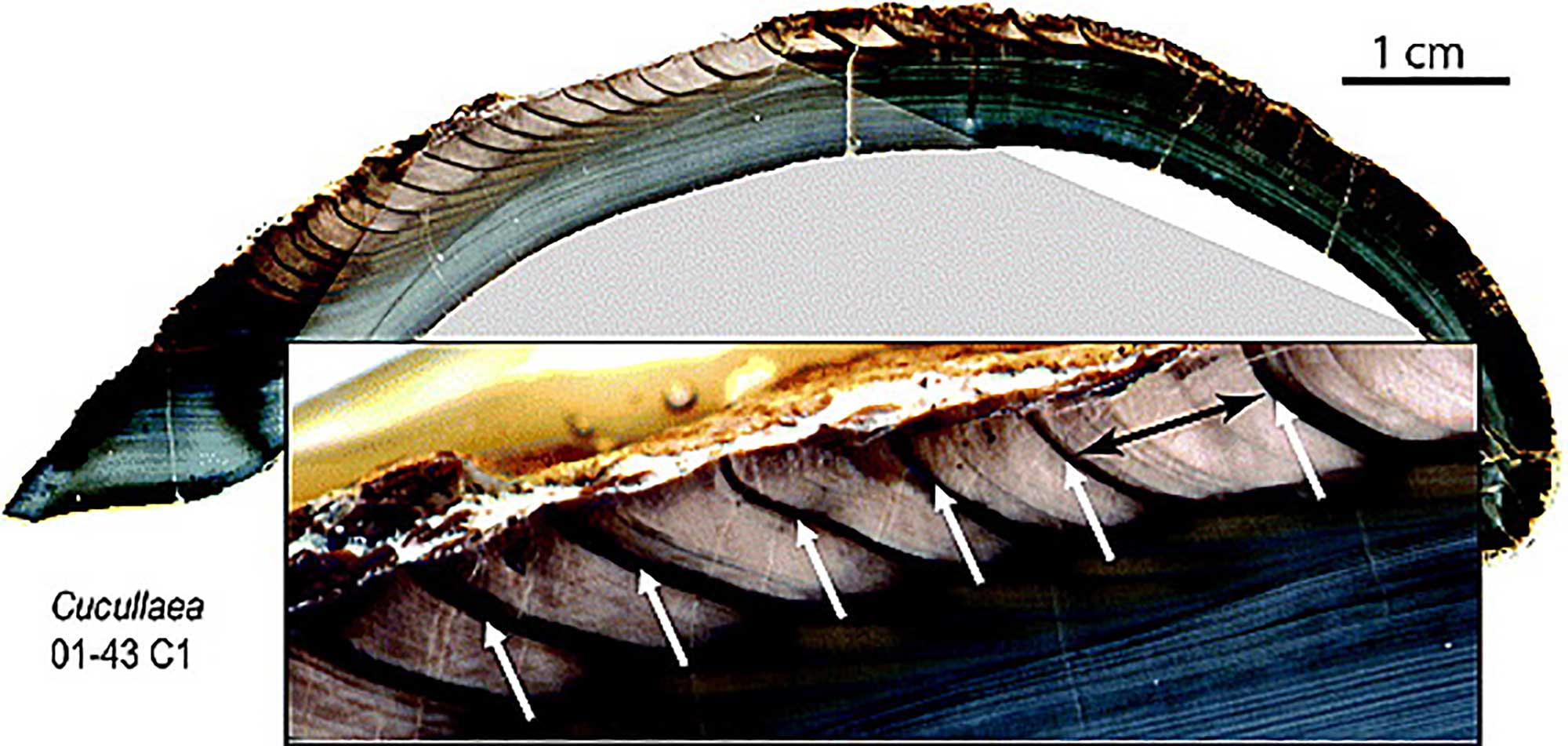
Cross-section through the shell of Cucullaea raea (Superfamily Arcoidea; Eocene, Seymour Island, Antarctica). This specimen shows increments of annual growth. Magnified view shows annual growth patterns (one year spanned by black arrow); the dark bands identified by white arrows represent the periods of slowest growth, in this case, summer. Image from Ivany et al., 2011 (fig. 2) in Geophysical Research Letters; image provided courtesy of Linda Ivany.
Although these growth lines look rather like tree rings, they do not necessarily reflect years of growth (see Sclerochronology, below).
The bivalve shell is usually composed of several layers of crystals of calcium carbonate (CaCO3).
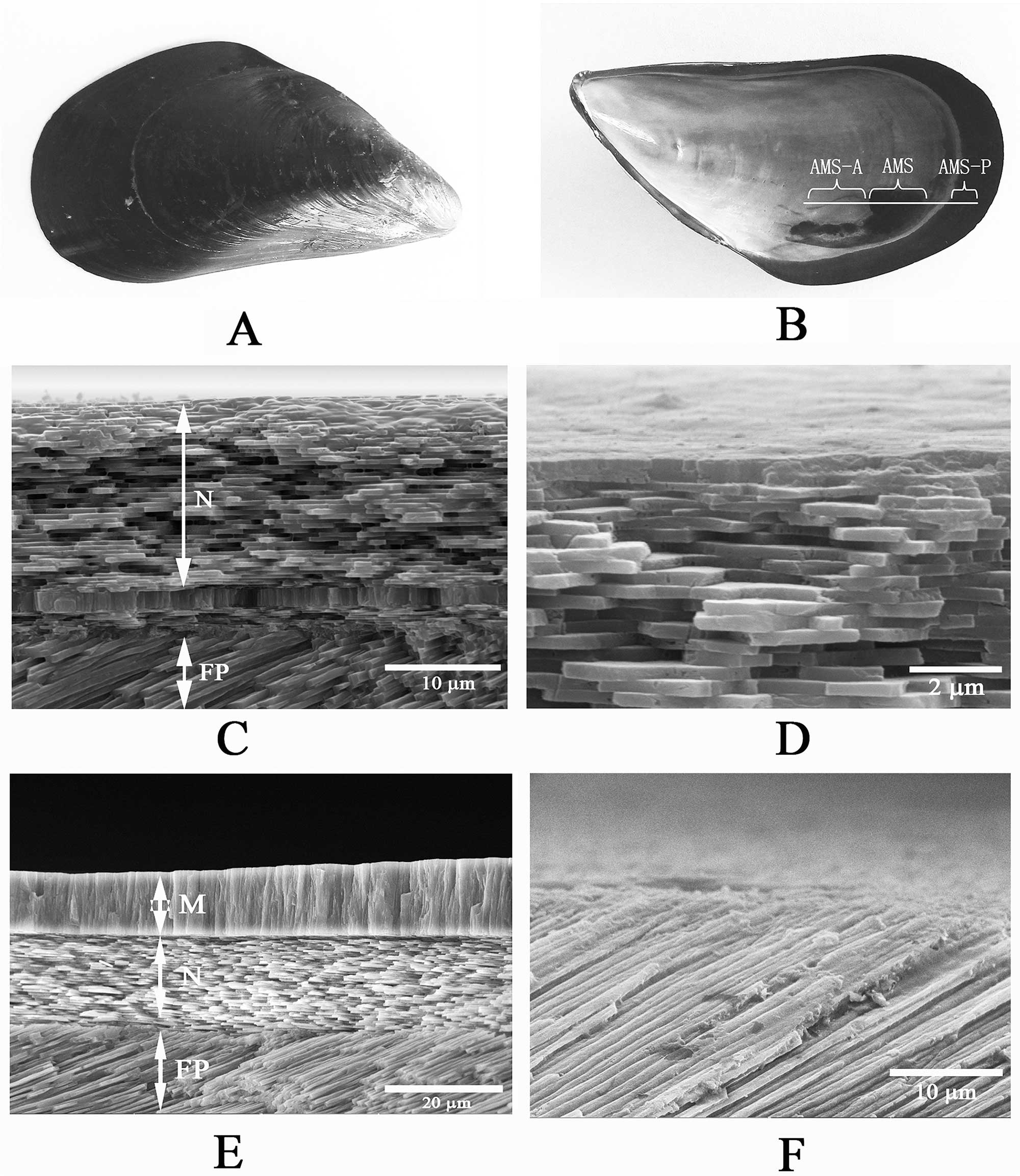
Composition is group-specific, but the major categories include: 1) nacreous layers (nacre or mother-of-pearl), always innermost, composed of tablet-shaped crystals of aragonite; 2) crossed-lamellar layers, the most common type, composed of crystals of aragonite obliquely positioned at angles to one another; 3) prismatic layers of columnar-oriented crystals of calcite or aragonite; 4) foliate layers, composed of sheet-like crystals of calcite, similar to nacre but differing in composition; and 5) homogenous layers of granular calcite or aragonite, lacking obvious crystalline structure. Detailed examination has identified many subgroups of these five major types. Holding the crystals together is organic matrix, composed of polysaccharide and protein (kind of like “mortar” to the crystal “bricks”), that also gives the shell its color; the crystals themselves are colorless. The shell of Mercenaria has two crossed-lamellar layers and an outermost prismatic layer. Exterior to these crystal layers is a usually uncalcified, thick or thin layer of organic material (the periostracum (see below; not well-developed in Mercenaria).

Periostracum on a live individual of the Banded Ark photographed in a tide pool at Mullaway Headland, New South Wales, Australia. Image by Harry Rose (flickr; Creative Commons Attribution 2.0 Generic license; label identifying periostracum added).
The periostracum provides protection against abrasion, and/or camouflage by attached sponges, algae, and other epibionts.
The ligament is considered a partly organic component of the shell (see image above under "Ligament"). Along with the hinge teeth, the ligament forms the hinge system that holds the two shell valves together. Usually brown or black in color, the ligament includes two distinct components: an inner portion that is partly mineralized with aragonite fibers, and an outer organic region composed of fibrous glycoprotein (conchiolin).

The resilium (internal ligament) of a scallop shell. Image by "Shellnut" (Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license; image cropped and label added).
The ligament can be external or internal (in which case it is called a resilium, with a structure for attachment called a resilifer). The ligament is not normally preserved in fossils, although fossil ligaments are known in exceptional circumstances from as old as the Carboniferous. More typically, clues about the ligament of fossil bivalves come from structures of the shell around the ligament, such as fossettes, nymphs, resilifers, etc.
Sclerochronology
Fossil and modern bivalve shells can yield information about their chronological age at death through the methods of sclerochronology.
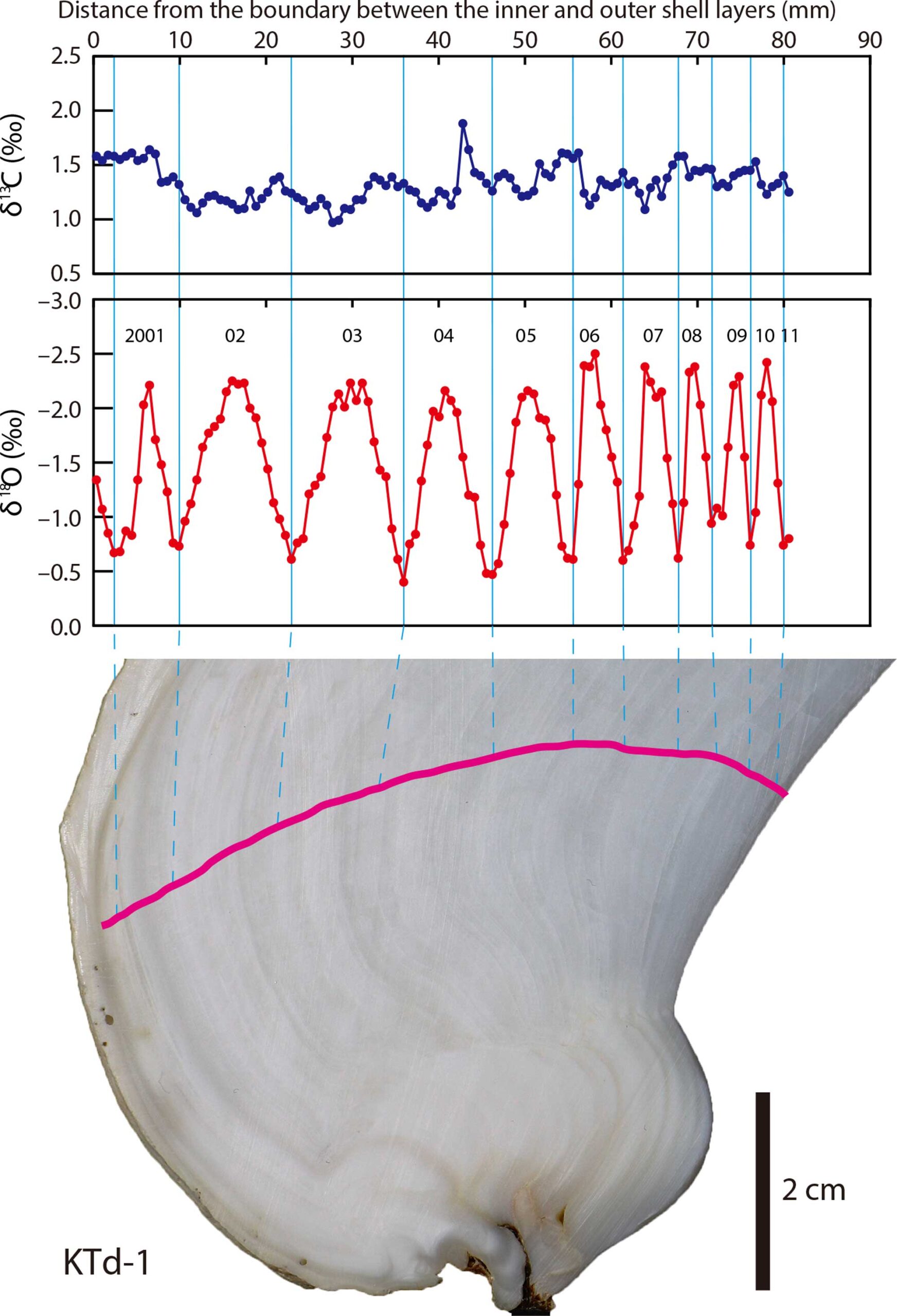
The top panel shows the carbon isotopes accumulated in the shell of the Tridacna clam. The middle panel shows the oxygen isotope profile, with each peak labelled for the year when the clam was alive. This clam was at least ten years old. The bottom image shows where the clam shell was sampled with a pink line. Notice that this pink line is perpendicular to the feint lines of the shell, which show grow within each year. Published caption: "δ13Cshell and δ18Oshell profiles of a Tridacna derasa shell (KTd-1). Pink line indicates the sampling transect." Figure from Yamanashi et al. (2016) in PLoS ONE; Creative Commons Attribution 4.0 International license.
This technique uses the temperature-dependence of the ratio between two stable (i.e., non-radioactive) isotopes of oxygen when it is incorporated into the calcium carbonate (CaCO3) of the bivalve shell. CaCO3 precipitated in warmer water is relatively high in 16O and depleted in 18O (“isotopically light”); CaCO3 precipitated in cooler water is relatively high in 18O and depleted in 16O (“isotopically heavy”). By taking samples across the shell, a profile of isotopic measurements can determine how many annual temperature cycles the bivalve lived through, and therefore how many years old it was when it died. This allows investigation of a wide variety of topics in extinct species, from growth rates to heterochrony in evolution. Learn more about biogeochemical analysis of fossil samples in the Paleoecology chapter of the Digital Encyclopedia of Ancient Life.
Soft Anatomy
Many species of animals have lost body parts during the course of their evolution. One of the main traits of all bivalves is the absence of a head and its associated organs such as eyes, head tentacles, and mouthparts. Scientists believe that early ancestors of bivalves had heads, like their close relatives the snails, but that bivalves have lost the feature.
The Mantle
The mantle covers the body and secretes the shell. The mantle is a complex sheet of tissue consisting of soft exterior membranes that secrete the shell and usually form a cavity – the mantle cavity – that encloses the gills and other organs, and which produces and enlarges the shell by secreting new shell material at the free edge. The mantle contains pallial muscles, which retract its edge and control the flow of water into and out of the mantle cavity. The mantle is comprised of two or three layers or folds.
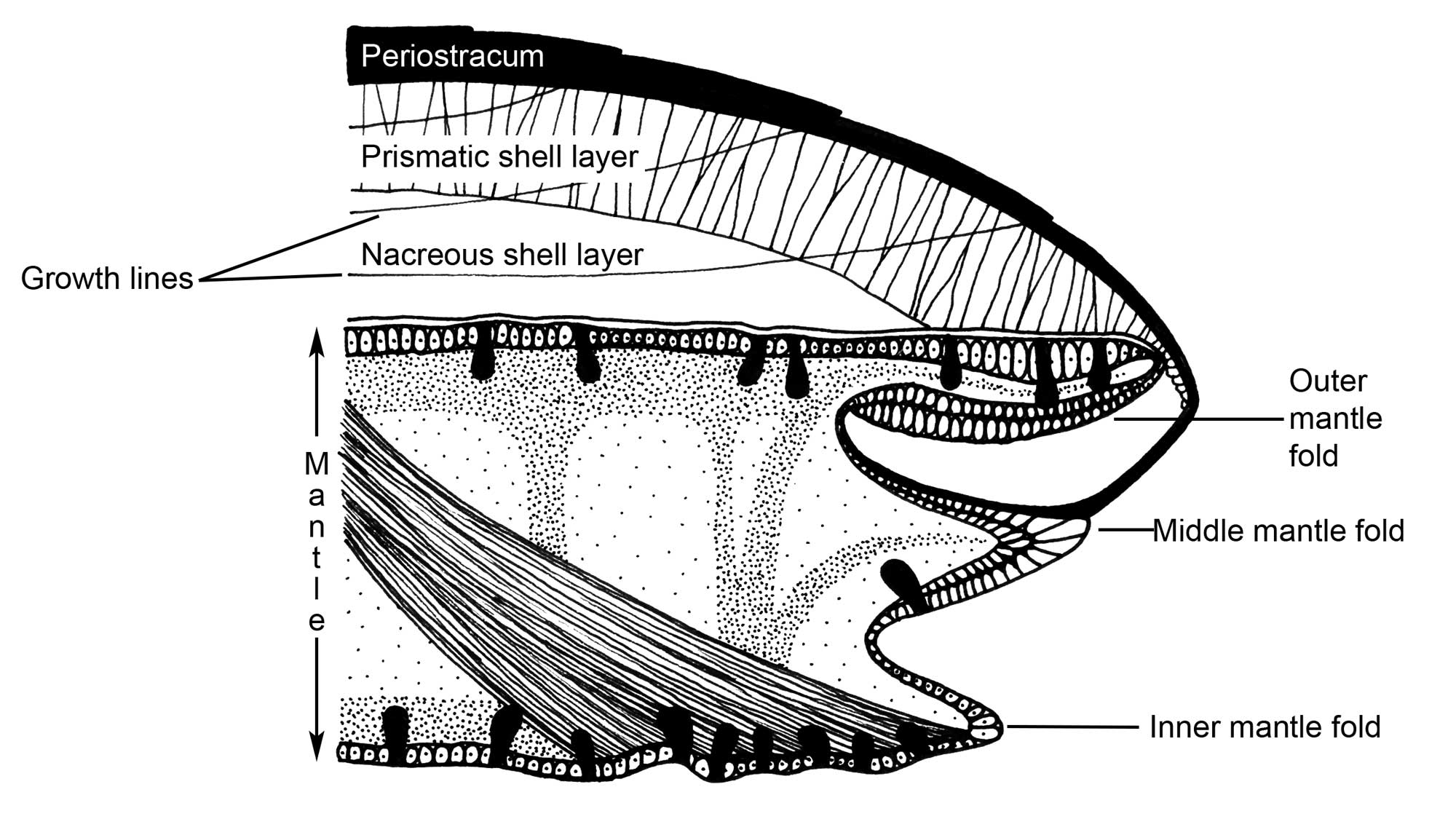
Cross-section through the edge of the mantle of a typical bivalve, showing the three folds of the mantle, the outer of which secretes the shell. (Drawing by Christi Sobel; modified from Barnes, 1980.)
The outer fold of the mantle secretes the shell, with its inner surface secreting periostracum and its outer surface secreting the calcareous layers. The inner fold is usually the thickest and is muscular. The middle fold is sensory, which is particularly important for bivalves, given their lack of a head. For example, this is where the tentacles and eyes of scallops are located.

The bay scallop Argopecten irradians. The eyes (blue dots) and tentacles are positioned on the mantle. Image by "Mayscallop" (Wikimedia Commons; public domain).
Remarkably, the mantles of some freshwater mussels are modified to resemble small fish. These adaptations serve as lures to attract real fish, which are hosts for the bivalve's parasitic larvae.
"Natural Fish Lure | Lampsilis Mussel and Bass" by Michael Backus (YouTube).
Siphons
The mantle also usually forms the siphons, usually through fusion of the edges of the mantle, although some species make siphons from mucus tubes.

Siphons in several burrowing infaunal modern species. Left: Dissected Mercenaria mercenaria, with the siphon held out in life position; photograph by Warren D. Allmon. Middle: The geoduck Panopea sp., whose siphon is so large that it cannot be fully retracted into the shell; image by The Evergreen State College (Wikimedia Commons; Creative Commons Attribution 4.0 International license). Right: Ruditapes philippinarum with separate incurrent and excurrent siphons extended; image by Stefan Didam (Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
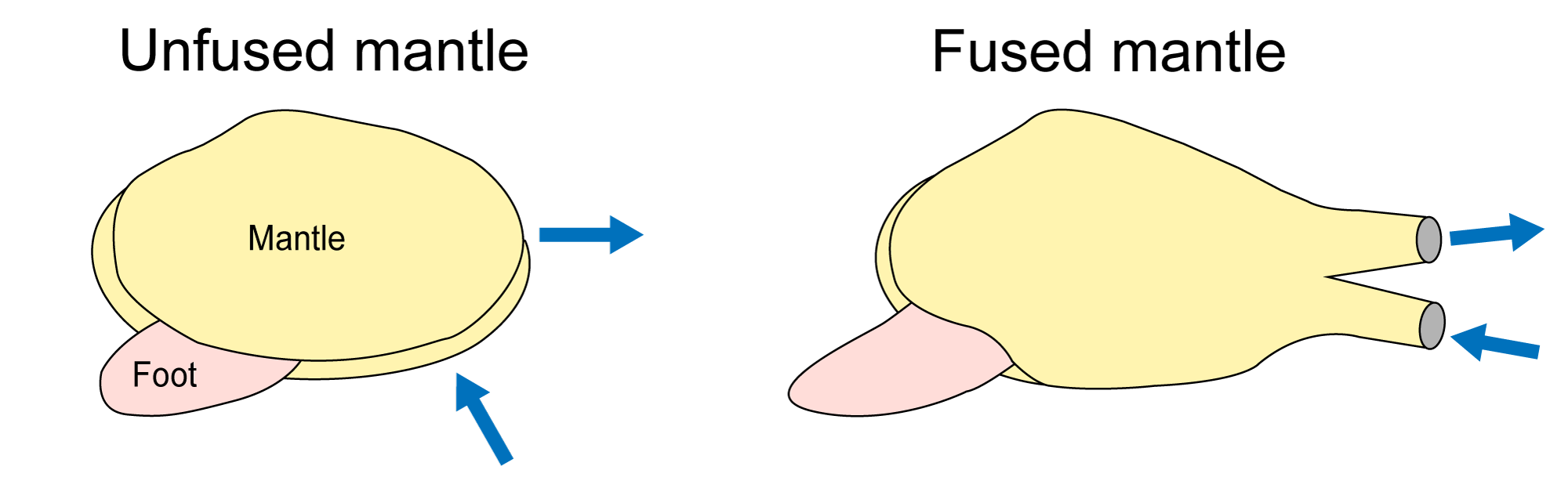
Diagrams showing how mantle fusion forms siphons in many infaunal bivalves (shell removed). Blue arrows indicate movement of water. Left: Bivalve with completely unfused mantle edge. Right: Bivalve with mantle edge fused to form paired siphons. Image by Jonathan R. Hendricks (Redrawn from Pojeta, 1987).
"Visualization of Freshwater Mussel Water Movement from In-current Siphon to Ex-current Siphon" by Jared McGovern (YouTube).
The incurrent siphon brings water and suspended food into the mantle cavity, and the excurrent siphon carries out water and waste. The two siphonal tubes can be separate or fused together. Bivalve siphons are attached to the shell by siphonal retractor muscles, which are enlarged parts of the pallial muscles. Dorsally (toward the hinge) the mantle is fused to the visceral mass and forms its integument; ventrally (toward the commissure) the mantle surrounds the open space of the mantle cavity. The mantle is attached to the shell ventrally along the pallial line which is generally parallel to the commissure (see images above). In species with a large siphon the pallial line shows an embayment (the pallial sinus) which allows for connection of larger retractor muscles. The depth of the pallial line is a good indicator of the size of the siphons (and therefore the burrowing ability) in bivalves, which can be very useful for reconstructing the habits of fossil forms (see section on Bivalve Ecology and Paleoecology).
Foot and Byssus
Most bivalves have a large foot, which is a muscular and expandable structure in the middle of the mantle cavity. The foot primarily functions as a burrowing organ in infaunal species. It can also function as an escape organ for some epifaunal forms (e.g., cockles, Cardium spp., family Cardiidae).
"Clam Digs into Sand" by DailyPicksandFlicks (YouTube).
"Cockle Jumping" by Fritz Rivera (YouTube). Video shows a cockle using its foot to escape a sea star predator.
Digging happens by pumping blood into the foot which expands the end and acts as an anchor; pedal retractor muscles then pull the bivalve into the substrate. The byssus is a set of elastic or calcified fibers secreted by a gland in the foot, used to anchor the bivalve to a hard substrate. Most bivalves have a byssus as larvae, and many lose it as adults.
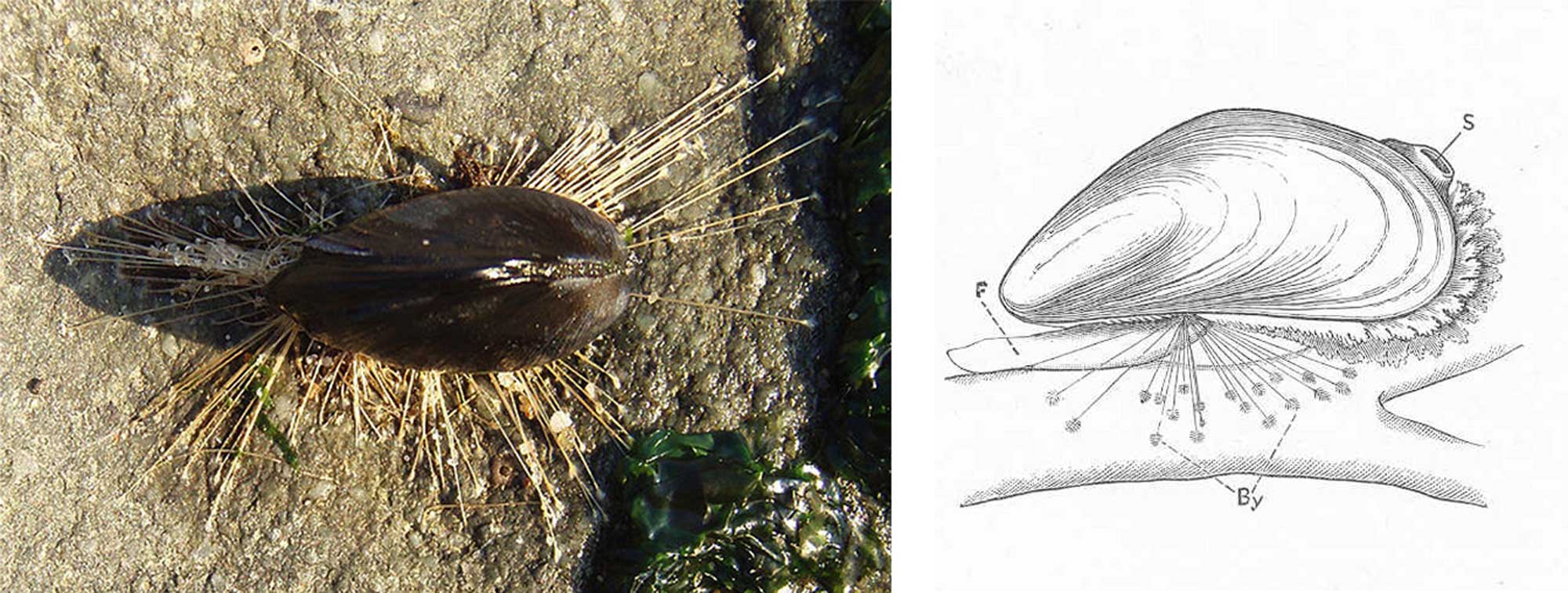
Photograph (left) and drawing (right) of a blue mussel (Mytilus edulis) secured to a rock by its byssal fibers. Left image by Brocken Inaglory (Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license). Right image from Cooke et al. (1895; Molluscs) (Wikimedia Commons; public domain).
Gills
All bivalves have gills (also called ctenidia), which they use for respiration; most species also use them for feeding.

Four types of bivalve gills: protobranch, filibranch, eulamellibranch, and septibranch (the latter three are all forms of autobranch gills). Drawings by Christi Sobel, based on Beerbower (1968) and Ponder et al. (2020.)
All bivalves have a single pair of gills, each of which is composed of a curtain of filaments and attached dorsally within the mantle cavity. The filaments are covered with cilia, whose beating in unison creates currents that move water over the gills. Gill structure is the morphological basis for dividing bivalves into two major groups: the protobranchs and autobranchs (see section on Bivalve Phylogeny and Classification).
Protobranch gills are typically rather small and simple, and used primarily for respiration. Autobranch gills are much longer and usually folded in a W-shape, forming ascending and descending branches on each side of the foot – each pair called a demibranch (see image above). Autobranch gills are usually used for suspension feeding in addition to respiration. Carnivorous bivalves (e.g., Poromyidae) have septibranch gills, comprised of a muscular septum (for sucking small prey into the mantle cavity) with small windows (ostia) containing respiratory gill filaments.
The mineralized shells of ancient bivalves have a superb fossil record, but, their soft parts are virtually unknown. An exception is provided by several remarkably preserved specimens of Laevitrigonia gibbosa from the Jurassic of England, which show evidence of gills and other soft parts.

Laevitrigonia gibbosa from the Upper Jurassic of England (ANSP 78266). This and two other associated specimens were first described in 1848, and are the first specimens of fossil mollusks—and apparently the first fossils of any sort—found to have preserved most of their soft anatomy. See Torrens et al. (2000) for more information. Photo by Chandler Olson, courtesy of Kathryn Estes-Smargiassi, Academy of Natural Sciences of Drexel University.
Gut
The gut or digestive system in bivalves runs from mouth to anus, including labial palps surrounding the mouth for initial particle sorting. The stomach is usually large, and more complex in autobranchs than protobranchs, including sorting areas and ducts to the digestive gland where useable food particles are digested.
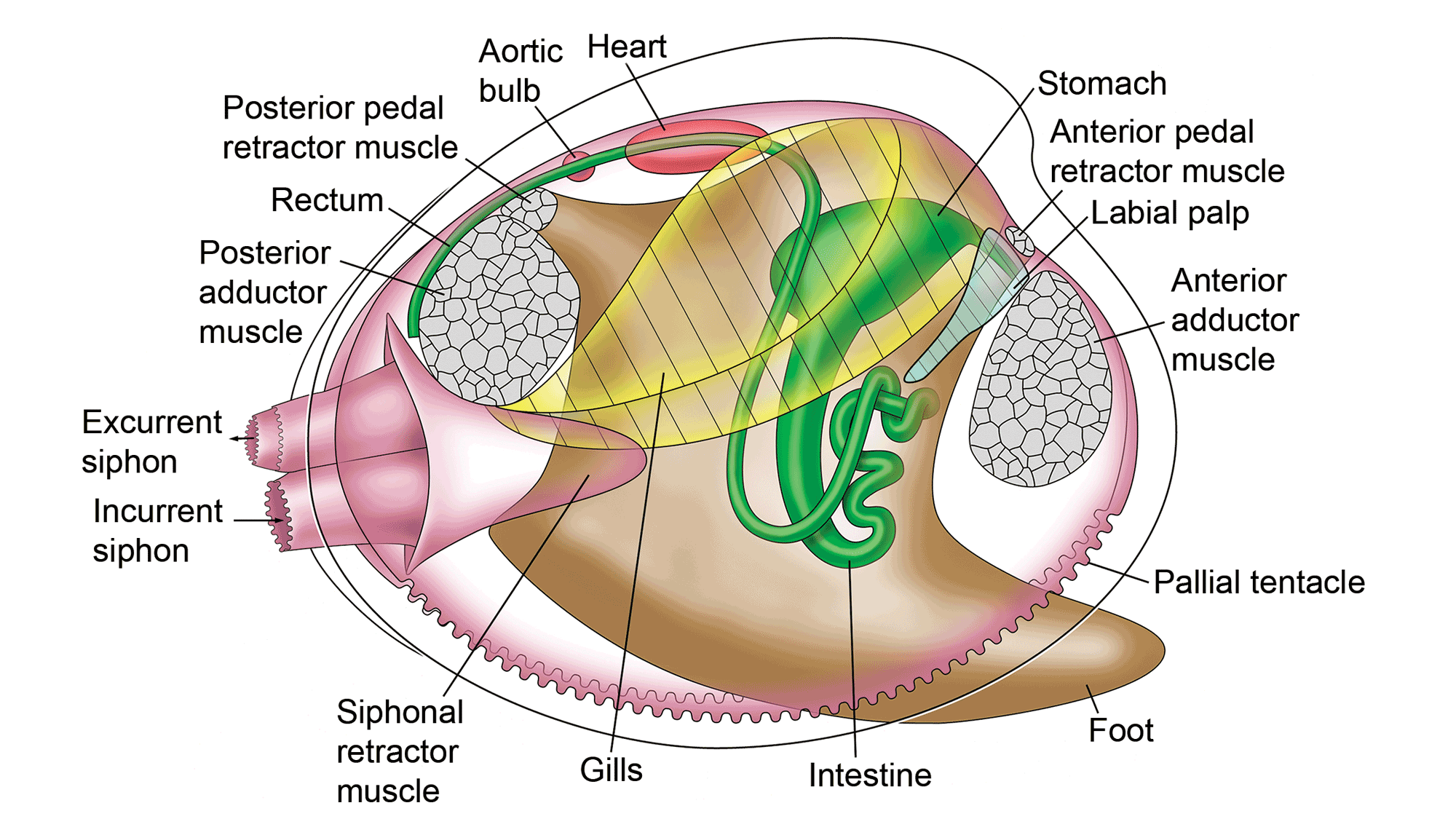
Soft anatomy of the hard-shelled clam, Mercenaria mercenaria. (Original artwork by Paula Mikkelsen and Lisa Kanellos, Field Museum of Natural History; from Mikkelsen and Bieler (2007), courtesy of Princeton University Press.)
In most autobranch species, it contains a crystalline style – a proteinaceous rod-shaped structure that rotates, stirring and macerating the stomach contents and releasing digestive enzymes. (Suspension-feeding gastropods, such as turritellids, also have a crystalline style.) Waste material exits from the stomach via the intestine, a usually long and coiled tube that extends ventrally into the visceral mass and dorsal part of the foot. The rectum runs across the posterior adductor muscle, opening near the excurrent aperture or siphon.
Muscles
One or two adductor muscles hold the shell tightly closed when they contract.
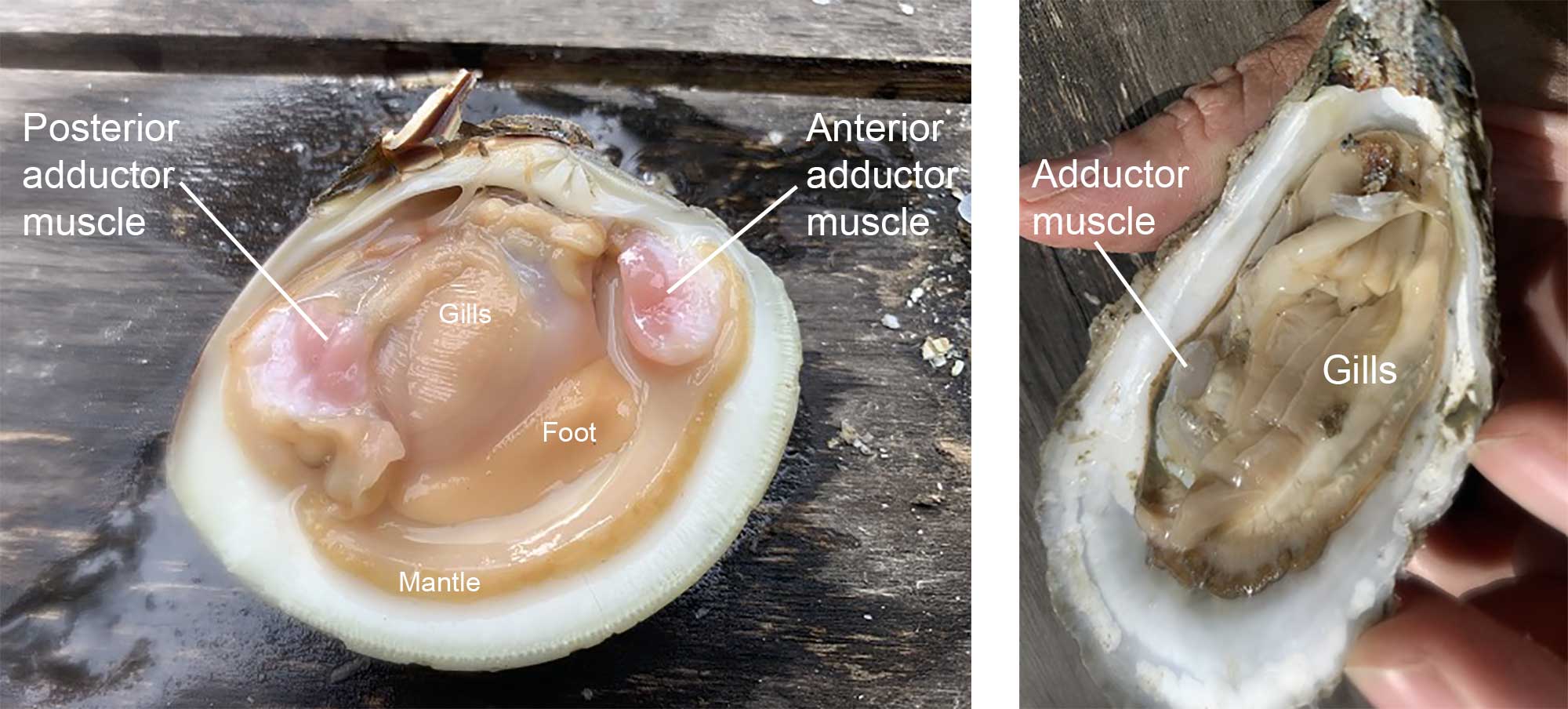
Dissected specimens of the clam Mercenaria mercenaria (left) and the oyster Crassostrea virginica (right). Mercenaria is isomyarian; Crassostrea is monomyarian. Photographs by Warren D. Allmon.
Species with two approximately equal-sized adductor muscles are called isomyarian; those with two unequal muscles are anisomyarian; those with only one muscle, including oysters and scallops, are monomyarian. When oysters are “shucked” for human consumption, the first step is to break the hinge, followed by a quick slice of the single adductor muscle. The adductor muscle is the part of the scallop that is consumed in the US; the entire scallop body is consumed elsewhere in the world.



