Chapter contents:
Class Gastropoda: Introduction, Morphology, Anatomy, and Life History
– 1. Phylogeny and Classification of Extant Gastropoda
– 2. Fossil Record of Gastropoda ←
– 3. References and Further Reading
– 4. Silly Snail Stuff
Image above: Specimens of the Devonian gastropod Bembexia from Delphi Falls, New York; collections of the Paleontological Research Institution.
Overview
Gastropods have left behind an extraordinary fossil record that documents a rich evolutionary history. They are distinctive among animals for their high taxonomic, ecological, and environmental diversity. The reasons for this evident “evolutionary success,” however, remain unclear. Their body plan is clearly a successful and evolutionarily flexible one, which has allowed them to invade numerous habitats and diversify into many different lineages and species; the only realm that they have not invaded is the air. Some of this diversification has clearly been “adaptive,” associated with differentiation into different habitats and ways of making a living. Some, however, is difficult to attribute to adaptation. Gastropods may also be unusual in having a high degree of “extinction resistance.” That is, once they have originated, gastropod taxa tend to persist for a long time, allowing the group as a whole to build up diversity. It has also been suggested that heterochrony, or change in developmental timing in evolution, has been an important process in gastropod evolution, allowing for rapid and dramatic phenotypic changes.
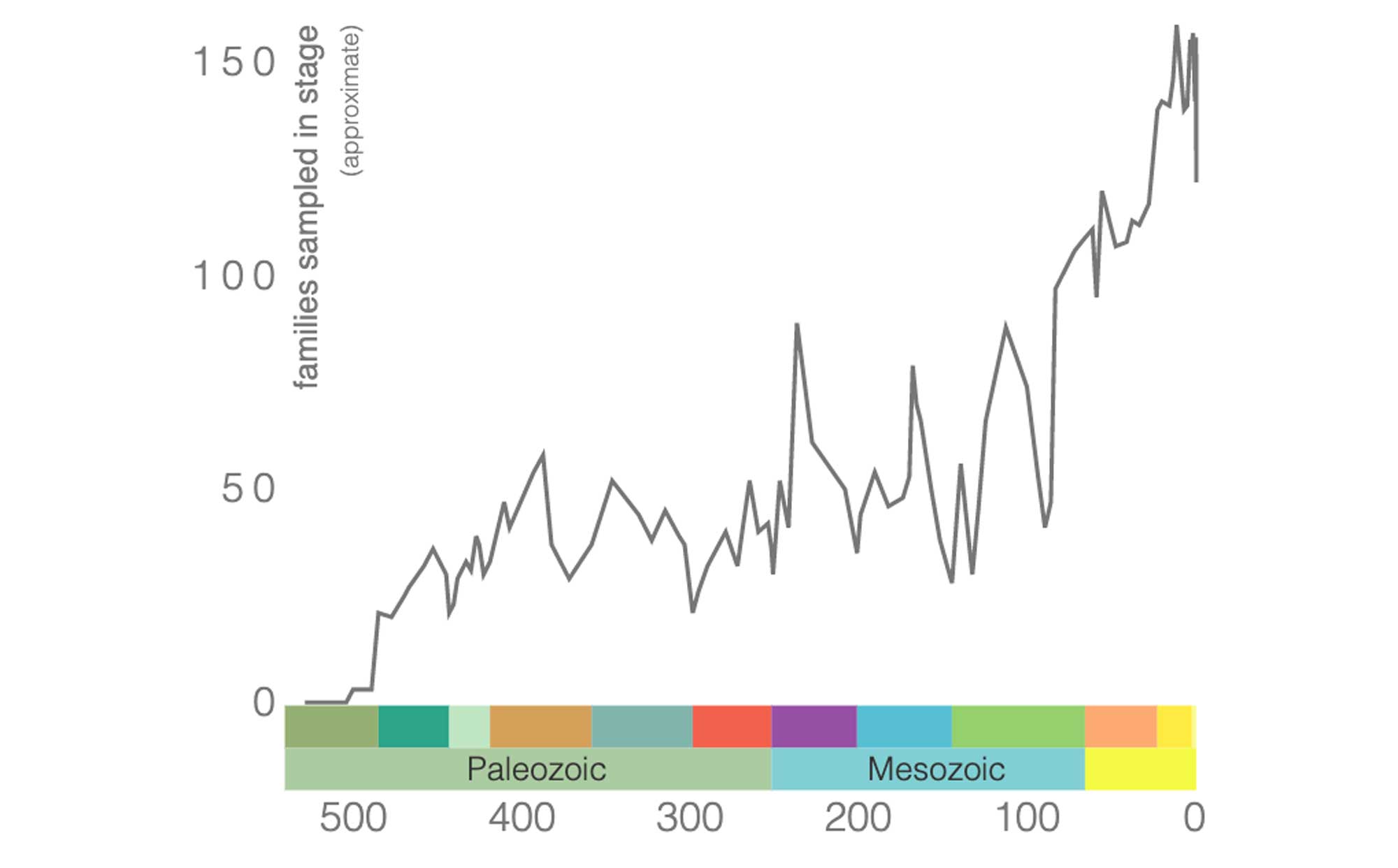
The graph below illustrates broad pattern of gastropod diversity over the span of the Phanerozoic Eon at the family level. These data were downloaded from the Paleobiology Database Navigator. Note the broad overall trend of gastropod diversity increasing over time.
Quick links to time intervals:
Gastropods During the Paleozoic Era
Overview
Gastropods occur widely if not always abundantly in Paleozoic rocks and made their first appearance in the Lower Cambrian. Determining the relationships of Paleozoic gastropod clades to one another, as well as to extant groups, is a challenging endeavor, most especially because of their paucity of shell characters, limiting their inclusion in phylogenetic analyses. Even in cases where it is possible to tease out characters and character states, morphological overlap and convergence obscure the differences between major fossil gastropod groups. Thus, most Paleozoic fossils lack obvious morphological features that might link them to extant clades.
The exception to this is characters of the larval shell (protoconch), which appear to be reliable indicators of phylogenetic relationship in Paleozoic gastropods (see examples below). Unfortunately, most Paleozoic fossil gastropods do not have well-preserved protoconchs. When they are preserved, however, they may provide connections to extant clades. The protoconchs of major extant gastropod clades exhibit the following characteristics:
- Caenogastropoda: more than one whorl; orthostrophic (dextral); well-separated whorls
- Vetigastropoda: about one whorl
- Neritimorpha: orthostrophic, but broader and more planispiral than caenogastropods
- Heterobranchia: heterostrophic (sinistral)
The presence or absence of such protoconch features in Paleozoic gastropods may help to evaluate their relationships (if any) to extant clades.
Using the Paleozoic fossil record to determine the early evolutionary relationships between the major living groups is particularly challenging. For example, taxa clearly identifiable as neritimorphs and caenogastropods are present in the Devonian. The two groups share planktotrophy and both have homeostrophic protoconchs. This may indicate that the two groups shared a common ancestor in the Silurian. On the other hand, it is possible that Ordovician subulitiform taxa known as perunelomorpha are stem-group caenogastropods, in which case the last common ancestor of the living major groups is Ordovician or even older. Discovery of well-preserved pre-Devonian protoconchs would shed light on this question (Fryda et al., 2008).
Cambrian
It is difficult to determine what the oldest known gastropod is, because we cannot directly observe torsion on fossils; it is therefore difficult to demonstrate that a particular fossil is in fact a gastropod, as opposed to a different class of untorted mollusk. During the Early Cambrian, there were clearly many types of univalve mollusks, most of which were probably untorted, and therefore not gastropods. Others were stem-group gastropods, that is they were torted univalve mollusks, but not part of clades that include modern gastropod taxa. Examples include Helcionellaceans, Aldanella, and Pelagiella.
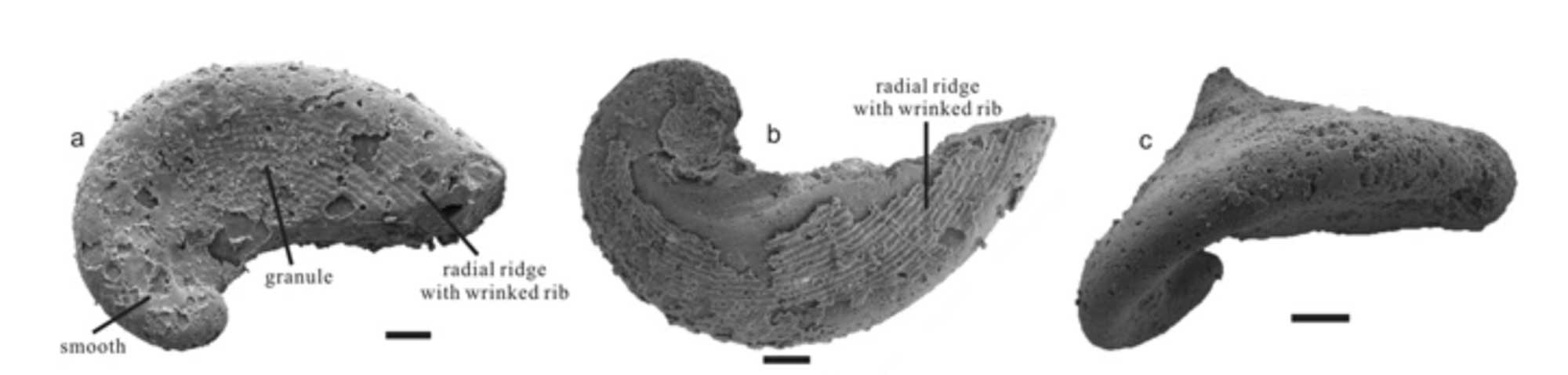
The Cambrian gastropod Pelagiella madianensis. Fig. 2 from Li et al. (2017). Original caption: "Morphology and ornaments of P. madianensis. (a) LC063505 (b) lc060159 (c) lc060326 shows the turbospiral, dextrally coiled shell with an open umbilicus and elongated aperture" (Li et al., 2017 in Scientific Reports; Creative Commons Attribution 4.0 International license).
Ordovician
Gastropods diversified rapidly and dramatically during the Ordovician, increasing in size, morphological disparity, and ecological diversity. Many new groups show their first appearance, including Euomphaloidea, Loxonematoidea, Lophospiroidea, Subulitoidea, Platyceratidae, and Macluritoidea (see examples below). Subulitoids may have included the first truly predatory gastropods, and macluritoids included sedentary suspension feeders.

Large specimens of Ordovician Maclurites sp. preserved on a bedding plane at Crown Point, Lake Champlain, New York. Photograph by Warren D. Allmon.
Fossil protoconchs suggest that the Ordovician also probably saw the origin of planktotrophic larvae among gastropods, as indicated by the implying an increase in the planktonic food supply in the world’s oceans.
Gastropod diversity dropped significantly at the end of the Ordovician, with at least 38 genera disappearing during the Ashgillian stage of the late Ordovician (Frýda, 2004).
Remainder of Paleozoic
The Silurian was a period of increasing gastropod diversity in most gastropod clades, as part of the recovery from the end-Ordovician mass extinction. Species with high spires became more common, as did taxa with slits at their apertures (Fryda et al 2008: p. 261). Platyceratids also diversified at this time. (Given their apparent specialization on parasitizing echinoderms such as crinoids, this might qualify as a true “adaptive radiation.”)
During the Devonian Period, gastropod diversity increased in both new and existing groups. Taxa with planktotrophic larvae (such as caenogastropods) expanded, suggesting an increase in planktonic food supply. Protoconchs of these species became more tightly coiled, suggesting increased predation on planktonic larvae. The effects of the late-Devonian mass extinction on gastropods are not well-known, largely due to poor fossil preservation at this time. The fact that Devonian and post-Devonian gastropod faunas share many taxa suggests that the extinction was not dramatic.
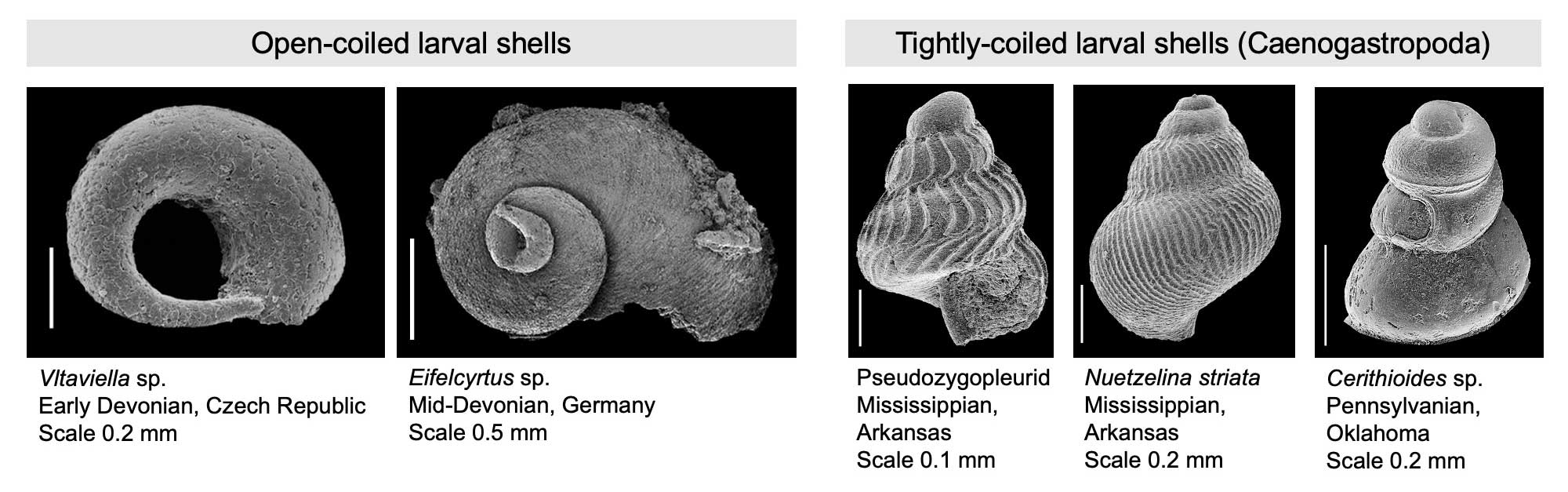
Early Paleozoic gastropod larval shells have open coils, whereas tightly-coiled larval shells characterize later Paleozoic species. Scanning electron microscope images of gastropod larval shells provided courtesy of Alexander Nützel. For additional information, see Nützel (2014 in Palaeontology).
Carboniferous and Permian gastropod faunas included abundant bellerophontids, euomphalids, slit-bearing vetigastropods, and naticopsids, and a continued expansion of high-spired caenogastropods, such as Pseudozygopleuridae, Orthonematidae, and Meekospiridae. The “subulitoid” family Soleniscidae originated in the Devonian and became widespread and diverse in the late Paleozoic. Both Pseudozygopleuridae and Soleniscidae may have been predators and so these changes may also qualify as genuine adaptive radiations. Cerithioids are definitely present in the Permian, if not before.
Some of the oldest possible land snails (e.g., Dawsonella meeki) occur in the Pennsylvanian rocks from Illinois (Kano et al., 2002), but the oldest definite terrestrial and freshwater gastropods are known from the Jurassic Period (Dayrat et al., 2011). Successful invasion of land and freshwater habitats required the evolution of internal fertilization, which apparently occurred independently in Neritimorpha, Caenogastropoda, and Heterobranchia.
Permian-Triassic Mass Extinction
The end-Permian mass extinction had a major and lasting impact on gastropods. In general, Paleozoic and post-Paleozoic gastropods differ markedly, and this is in large part due to the extinction. For example, Paleozoic gastropods mostly had two gills, were slow-moving suspension-feeders or herbivores, and frequently had little external shell ornamentation. Post-Paleozoic gastropods, in contrast, include more active, mobile forms, as well as more carnivores; most have a single gill, which allows for a more elongate aperture, the development of an inhalant siphon, and a more streamlined shell shape. In detail, however, the changes that accomplished this transition were complex. Global diversity of gastropods began to decline as much as 14 million years before the end of the Permian, with about 45% of genera becoming extinct at the very end (Erwin, 1990; 2006: p. 113; Erwin and Hua-Zhang 1996: p. 225).
A number of important Paleozoic groups became extinct at the end of the Permian, including euomphalids, pseudozygopleurids, orthonematids, and platyceratids. Other Paleozoic taxa trickled through, including bellerophontoids and soleniscids, the latter possibly including precursors of some Mesozoic caenogastropod clades. Other Paleozoic survivors were much less successful and eventually disappeared. The taxa that would come to dominate the Mesozoic did not fully take over until the Middle and Late Triassic. In the immediate aftermath of the Permian-Triassic extinction, marine gastropods were unusually small everywhere, possibly as a result of the dramatic change in food supply at the extinction event (Fraiser and Bottjer, 2004; Payne, 2005; but see Nützel, 2005).
Major Paleozoic Gastropod Groups
Some of the most abundant, diverse, and widespread Paleozoic gastropod groups are described below. Most or all of these groups may be polyphyletic.
Euomphaloidea
Lower Ordovician to Triassic. Usually disc-shaped (planispirally coiled). Early whorls often bearing septa. Presumed exhalent channel often present on apertural margin. Outer shell layer calcitic. May be vetigastropods. Examples: Amphiscapha, Trepospira.

Amphiscapha subrugosus from the Pennsylvanian Graham Fm. of Stevens County, Texas (PRI 70111). Image by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
Murchisonioidea
Lower Ordovician to Triassic. Usually high-spired, with sinus on apertural margin ending in short slit or notch generating selenizone. Examples: Murchisonia, Trochonema.

Trochonema becoitense from the Middle Ordovician Platteville Limestone of Illinois (PRI 70107). Image by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
Loxonematoidea
Middle Ordovician to Jurassic. Usually high-spired, lacking an umbilicus. Sinus usually present, deep to shallow, but without a notch at the apertural margin. Example: Loxonema.

Loxonema terebra from the Devonian of Tompkins County, New York (PRI 70110). Image by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
Lophospiroidea
Ordovician to Triassic. Shell form variable; spire height usually moderate. Median labral sinus on apertural margin, usually forming a median angulation and selenizone on outer shell surface. Example: Worthenia.
Subulitoidea
Ordovician to Triassic. Shell ovoid, with large body whorl and relatively short spire; aperture usually tapered on both ends. Most taxa in this group probably caenogastropods. Examples: Meekospira, Subulites, Soleniscus, Ianthinopsis, Procerithiopsis, Girtyspira, Labridens, etc.

Specimens of Meekospira peracuta from the Pennsylvanian Graham Fm. of Coleman County, Texas. Collections of the Paleontological Research Institution. Photograph by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
Platyceratidae
Ordovician to Permian. Usually low-spired, with large aperture. Frequently associated (and assumed to be parasitic) on crinoids. Outer shell layers thick and calcitic. Probably polyphyletic (see Frýda et al., 2009). Example: Platyceras.
Bellerophonts
Cambrian to Triassic. Planispirally coiled (bilaterally symmetrical), with large and frequently flared aperture. Prominent median sinus or slit and selenizone. Shell microstructure probably cross-lamellar. Based on protoconch and internal muscle scars, bellerophonts may not be a single clade; that is, they may be polyphyletic, and include both untorted (monoplacophoran-like) and torted (gastropod) forms, with the latter being stem-vetigastropods. Example: Bellerophon.
Macluritoidea
Upper Cambrian to Devonian. This distinctive group was especially common in the Ordovician. Their shells had a flattened shape, and they attained large sizes, in some cases reaching 26 cm (Novack-Gottshall and Burton, 2014). They apparently were stationary suspension feeders (which is an unusual mode of feeding for gastropods, though it has evolved independently multiple times). Careful examination of the cross-sectional shape of the shell in macluritids during their ontogeny reveals that they underwent a transition, from being a typical mobile snail when young, and switching to life as a sedentary suspension feeder when mature. Example: Maclurites.
Paragastropods
A number of Paleozoic helically coiled shells may or may not have been gastropods. Genera such as Onychochilus (Silurian), Kobayashiella, and Matherella (Cambrian) are sinistrally coiled and may have had untorted bodies. They have been referred to a separate molluscan class called Paragastropoda. (See Linsley and Kier, 1984; Ponder et al., 2020, v. 2, p. 42.)
Gastropods During the Mesozoic Era
The Triassic was a time of rapid radiation of most of the major groups of gastropods, including neritimorphs, caenogastropods, and heterobranchs. The oldest patellogastropod fossils come from the Triassic. By the end of the period, caenogastropods, especially the predatory neogastropods, began their rapid and enormous evolutionary diversification. The oldest known neogastropod may be the Lower Triassic genus Pseudotritonium (Nutzel, 2010).
The radiation of caenogastropods in general, and neogastropods in particular, is an important element of a major transformation of life in the world’s oceans that took place during the Jurassic and Cretaceous. This change, known as the Mesozoic Marine Revolution (MMR), was marked by increased levels of primary productivity—perhaps fueled by increased nutrients from volcanism and runoff from the land. This productivity fed more consumers such as herbivores, which in turn fed more predators. This increased level of predation led to an “arms race” between predator and prey. (See Vermeij, 1987, 1995; Harper, 2003).
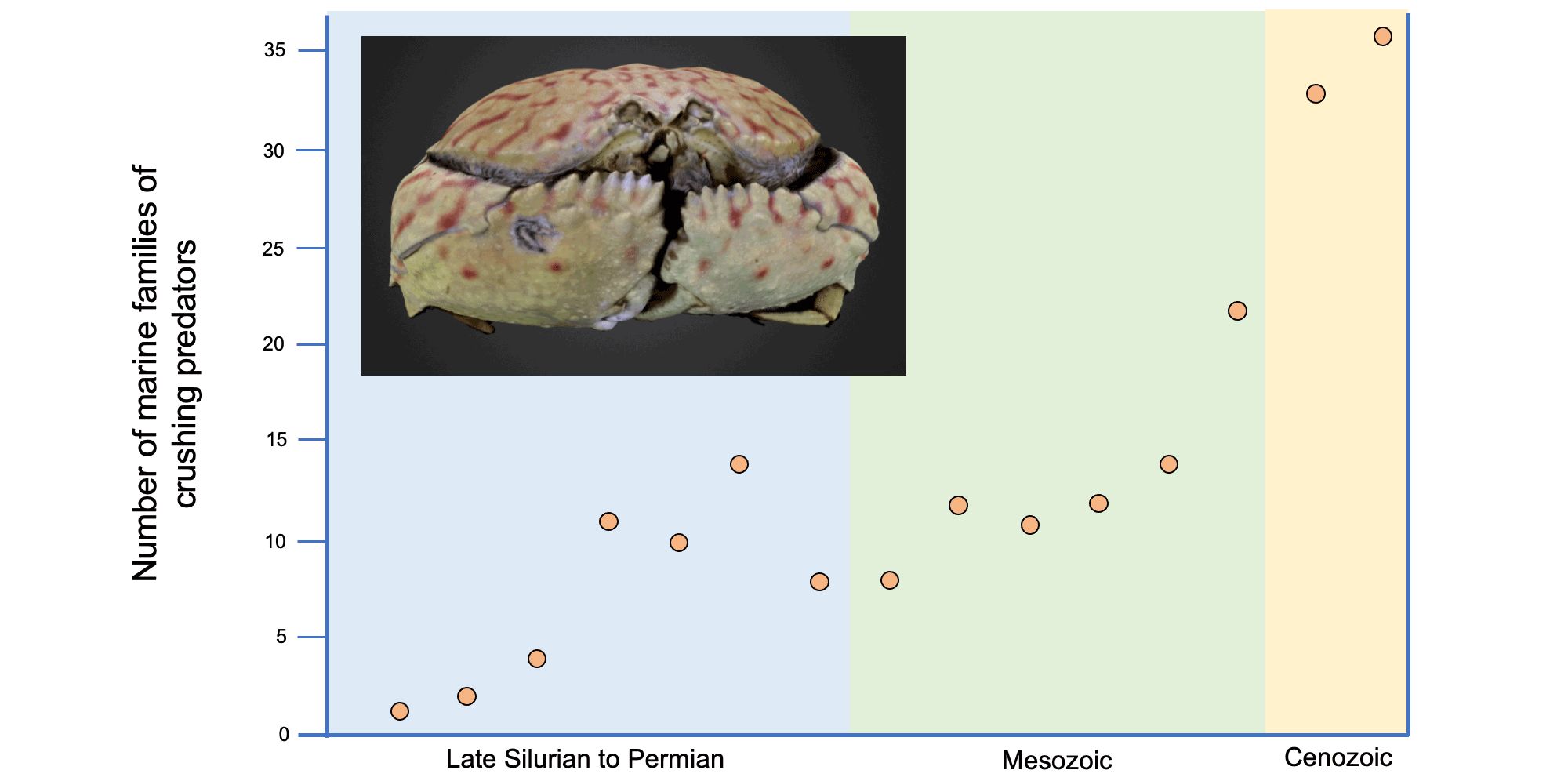
Image modified and reproduced from fig. 6.4 in Vermeij (1987). Original caption: "Number of marine families specialized for predation by shell breakage. Groups considered include eurypterid and crustacean arthropods, cephalopod molluscs, and vertebrates." Image by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
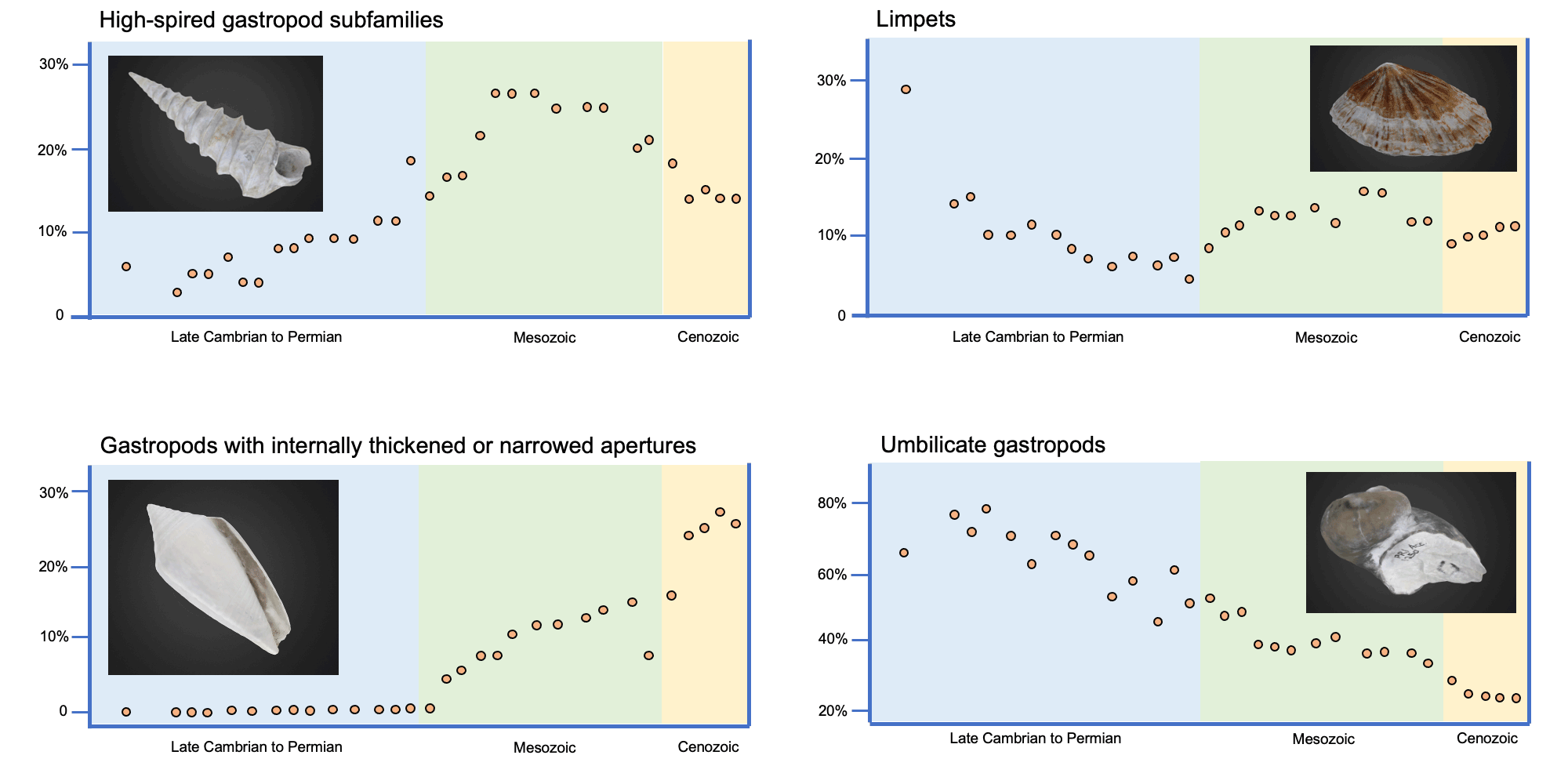
Image modified and reproduced from fig. 7.2 in Vermeij (1987). Original caption: "Subfamilial diversity of gastropods with various predation-related shell traits through the Phanerozoic." Traits include high-spired shells, limpet-shaped shells, shells with internally thickened of narrowed apertures, and umbilicate shells. Image by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
Mesozoic marine gastropod shells are more robust and ornamented than are their Paleozoic predecessors. Post-Triassic gastropods exhibit more sculptural features, such as spines, tubercles, ribs, varices, and apertural obstructions such as plications and teeth, which appear to have served a mechanically defensive role. Gastropods with umbilicate and planisprial shell shapes declined in diversity.
New predatory behaviors appeared in caenogastropods during the mid-late Mesozoic. Naticids (moon snails) and muricids (murex snails) started drilling holes in the shells of both bivalves and other gastropods. The intensity of this predation – and the diversity of all predatory neogastropods – increased dramatically in the Cretaceous.
In some respects, the gastropod fauna of the Cenozoic was established during the Cretaceous. For example, a number of modern families of neogastropods show their first appearance during the Late Cretaceous (Taylor et al., 1983), but there may also have been a number of other groups that appeared then and became extinct quickly thereafter (Bandel and Dockery, 2012).
An exception to the generally “modern” appearance of Cretaceous gastropod faunas is the nerineoids. The Nerineoidea were a group of large, diverse (> 90 genera), high-spired gastropods widely distributed in low-latitude shallow marine realms (especially carbonate environments) during Jurassic and Cretaceous times. They were frequently extremely abundant in environments in which they occur. They were likely slow-moving shell draggers and probably deposit and/or suspension feeders with a semi-infaunal mode of life, similar to living Turritellidae. They were distinctive, however, in having the space inside their shells limited by prominent wall protrusions or plaits, the function(s) of which have been much debated. Their protoconchs were apparently heterostrophic, and they are usually referred to Heterobranchia. Nerineoids became extinct at the end of the Cretaceous.
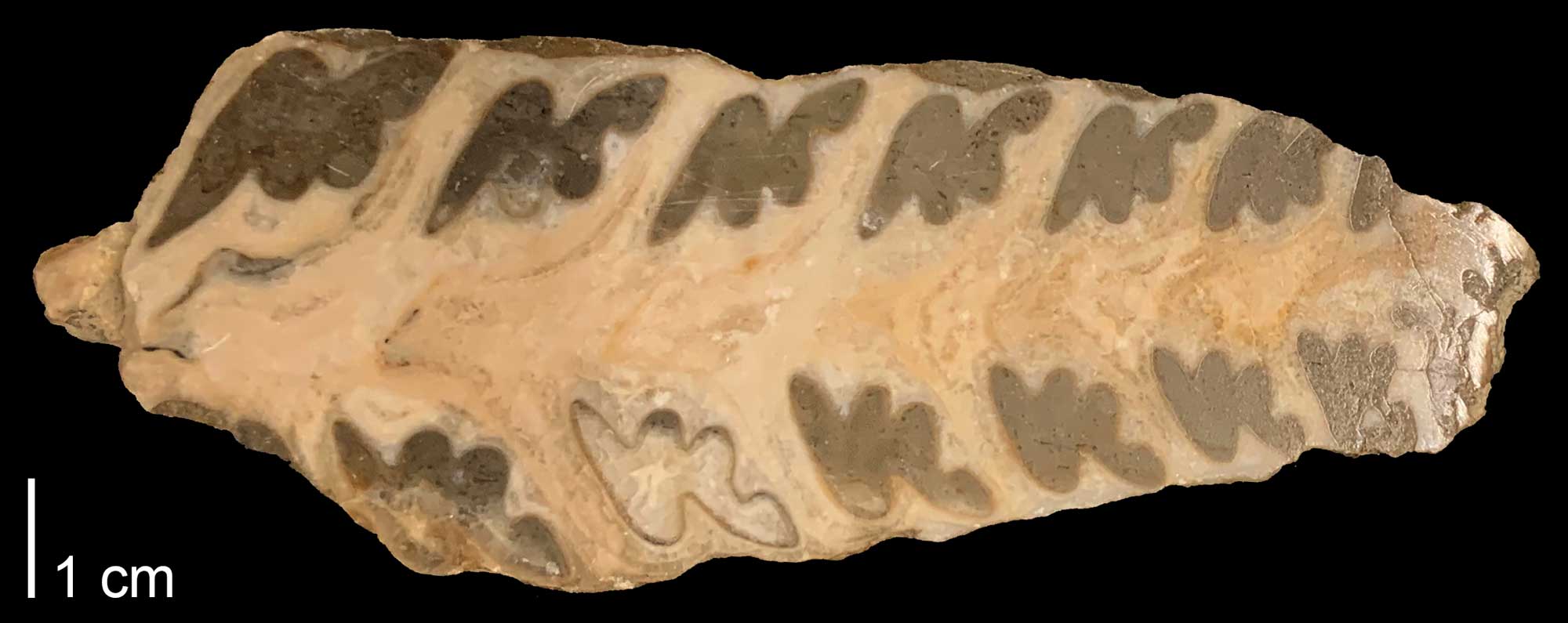
The nerineoid Simploptyrix pailletteana from the Upper Cretaceous Gosau Beds of Austria, cut in half to show interior plaits (PRI 104762). Specimen is from the collection of the Paleontological Research Institution, Ithaca, NY. Photograph by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.

Specimens of nerineoids (Nerinea?) from the Cretaceous of Syria; collections of the Paleontological Research Institution. Photograph by Jonathan R. Hendricks for the Digital Atlas of Ancient Life project.
The end-Cretaceous mass extinction had a smaller effect on gastropods than on many other animal groups. About 10% of families became extinct globally, including the distinctive, diverse, and abundant nerineoids. In some regions, local extinction at genus and species levels was much higher (for example, in the U.S. Gulf Coastal Plain), but these taxa belonged to groups that apparently survived in refugia elsewhere. A number of diverse groups of caenogastropods that had diversified during the Late Cretaceous continued to undergo major diversification in the Cenozoic.
Gastropods During the Cenozoic Era
Evolution of marine gastropods, especially caenogastropods, in the Cenozoic was in many ways a continuation of trends established in the Late Cretaceous. Neogastropods and cerithioids diversified rapidly in the Paleogene, with many modern genera, including Conus (cone snails), originating during this interval.
The Paleogene also saw the appearance of a group of pelagic heterobranchs known as pteropods, which became an important part of the plankton.
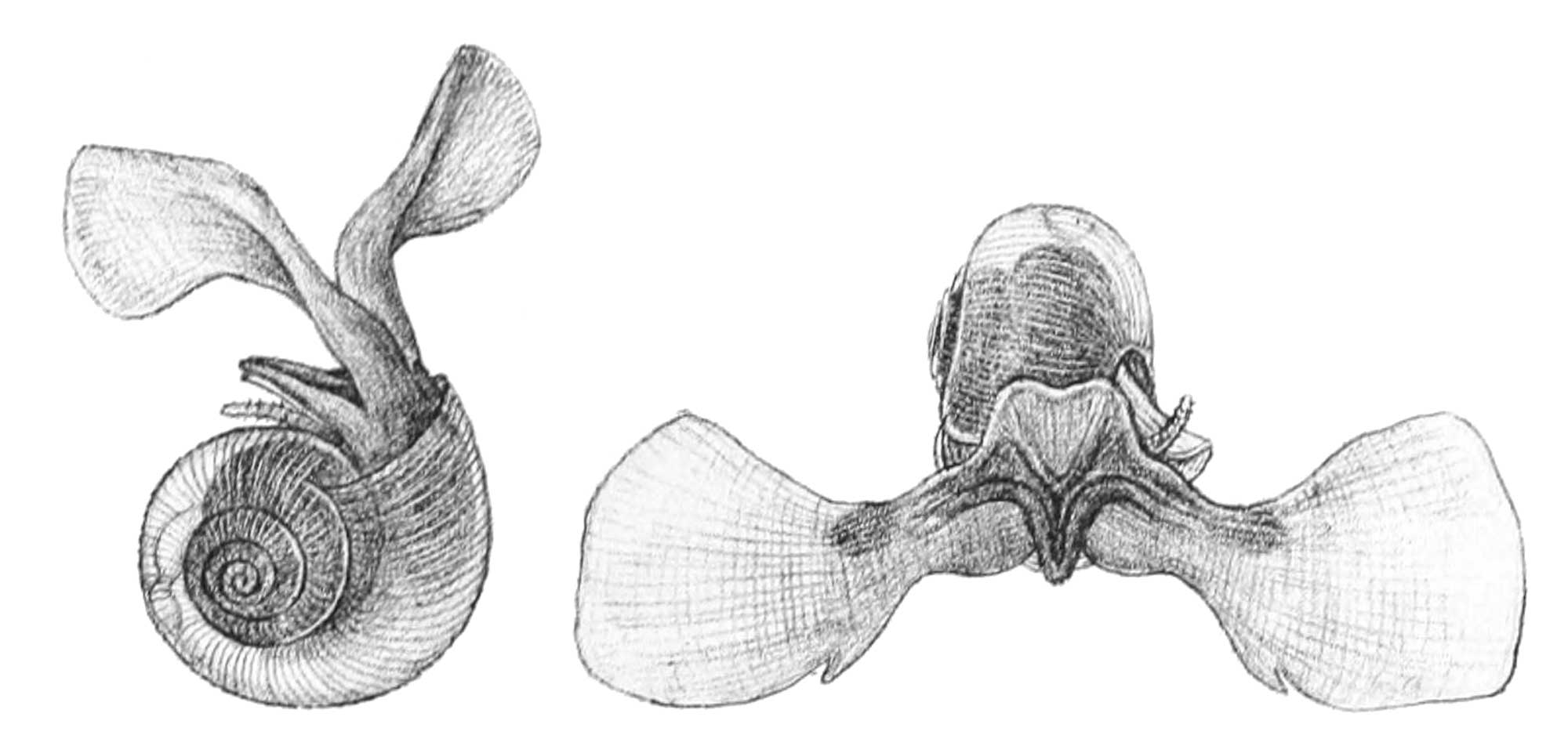
Drawings of the extant pteropod Limacina helicina by Georg Ossian Sars (1878; public domain).
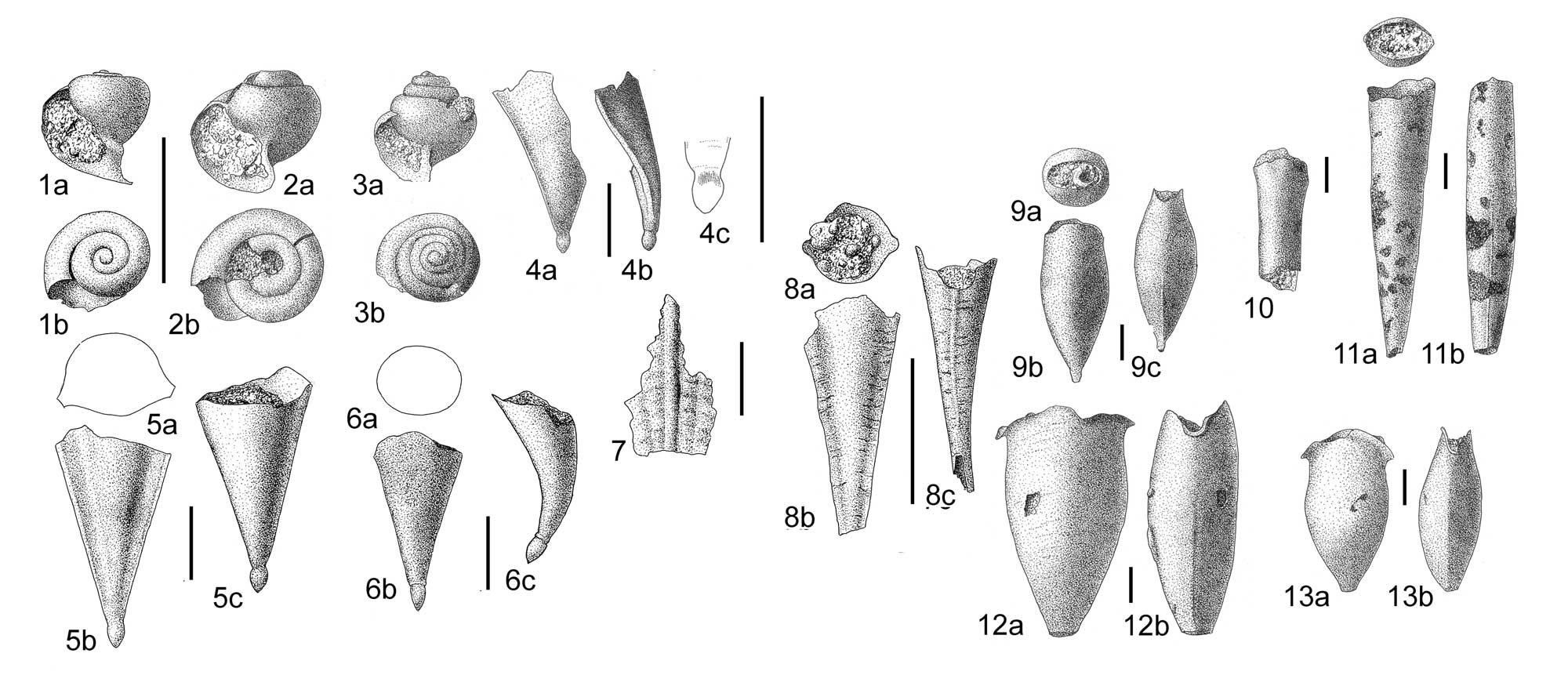
Examples of pteropod fossils from the Miocene Cantaure Formation of Venezuela. 1-3, Limacina spp.; 4-7, Clio spp.; 8, ?Diacria sp.; 9, Vaginella depressa; 10, 11, Vaginella lapugyensis; 12, 13, Vaginella grenadinarum. Image is plate 95 from Landau et al. (2016 in Bulletins of American Paleontology, copyright Paleontological Research Institution). Drawings by Arie W. Janssen. All scale bars equal 1 mm.
Changes in global climate and tectonics affected gastropod biogeography during the Cenozoic. The “closure” of the circum-equatorial Tethys seaway and cooling temperatures in the Oligocene and Miocene eventually destroyed shallow, warm marine habitats from the Mediterranean to India. Many gastropod taxa that had been globally distributed at the beginning of the Paleogene had much more restricted distributions by the late Neogene, frequently in the Indo-Pacific, which today has by far the highest diversity of marine gastropods.
Cenozoic gastropods, and the amazingly rich and diverse fossil record that they left behind in many places, have been the subjects of a number of important paleobiological studies.

Beds of Turritella mortoni in the Paleocene Aquia Formation, Bull Bluff, Potomac River, King George County, Virginia. Pick is approximately 1 m long. Photograph by Warren D. Allmon.
For example, the theory of punctuated equilibria was based in part on analysis of Pleistocene land snails of the genus Poecilozonites in Bermuda (Gould, 1969). Gould’s studies of another island land snail group, Cerion, contributed to his thinking on evolutionary constraint (Gould, 1984, 1989).

Poecilozonites bermudensis, one of the land snail species studied by Stephen Jay Gould in the original formulation of punctuated equilibria (Eldredge and Gould, 1972). Specimen is from the Pleistocene of Bermuda and is from the collections of the Paleontological Research Institution, Ithaca, New York (PRI 76831). Image by Jonathan R. Hendricks.
Heterochrony in Cenozoic fossil gastropods has been examined by a number of authors (e.g., Geary, 1988; Allmon, 1994; Dominici et al., 2020), as have speciation and adaptive radiation (e.g., Hansen, 1978; Nehm and Geary, 1994; Allmon and Smith, 2011; Glaubrecht, 2011; Neubauer et al., 2013).
Content usage
Usage of text and images created for DEAL: Text on this page was written by Warren D. Allmon or Jonathan R. Hendricks. Original written content created by Warren D. Allmon or Jonathan R. Hendricks for the Digital Encyclopedia of Ancient Life that appears on this page is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. Original images and diagrams created by Jonathan R. Hendricks or Warren D. Allmon are also licensed under Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Content sourced from other websites: Attribution, source webpage, and licensing information or terms of use are indicated for images sourced from other websites in the figure caption below the relevant image. See original sources for further details. Attribution and source webpage are indicated for embedded videos. See original sources for terms of use. Reproduction of an image or video on this page does not imply endorsement by the author, creator, source website, publisher, and/or copyright holder.
Adapted images. Images that have been adapted or remixed for DEAL (e.g., labelled images, multipanel figures) are governed by the terms of the original image license(s) covering attribution, general reuse, and commercial reuse. DEAL places no further restrictions above or beyond those of the original creator(s) and/or copyright holder(s) on adapted images, although we ask that you credit DEAL if reusing an adapted image from the DEAL website. Please note that some DEAL figures may only be reused with permission of the creator(s) or copyright holder(s) of the original images. Consult the individual image credits for further details.



