Chapter by:
Jaleigh Q. Pier and Jansen A. Smith, Paleontological Research Institution, Ithaca, New York.
An initial version of this chapter was first publicly shared on December 20, 2019; the complete chapter was first shared on July 24, 2020.
This chapter was last updated on July 24, 2020.
Chapter citation:
J.Q. Pier and J.A. Smith. 2019. Echinodermata. In: The Digital Encyclopedia of Ancient Life. https://digitalatlasofancientlife.org/learn/echinodermata
Chapter contents:
Echinodermata ←
–– 1. Exclusively Fossil Taxa
–– 2. Crinoidea
–– 3. Asteroidea
–– 4. Ophiuroidea
–– 5. Echinoidea
–– 6. Holothuroidea
Associated materials:
Virtual Teaching Collection of 3D photogrammetry models of Echinodermata fossils available here!
Above image: Recent preserved Echinodermata specimens. Image by Jonathan R. Hendricks of modern echinoderm specimens displayed at the Academy of Natural Science of Drexel University, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
Phylum Echinodermata Snapshot
- Living classes: Crinoidea, Asteroidea, Ophiuroidea, Echinoidea, Holothuroidea
- Exemplar extinct classes: Blastoidea, Ctenocystoidea, Diploporita, Edioasteroidea, Eocriniodea (as many as 28 exclusively fossil classes have been identified)
- Diversity: ~10,995 extant species, 30,325+ extinct species
- Ecology: exclusively marine (stenohaline), herbivore, carnivore, detritivore, filter feeder
- Key features of group: 5-part symmetry, stereom, endoskeleton of CaCO3 ossicles or plates, water vascular system with tube feet
- Fossil Record: Cambrian to Recent
Overview
“Among the echinoderms we find some of the most beautiful of all sea creatures." (Nichols, 1967, pg. 15)
The phylum Echinodermata represents the largest animal phylum of exclusively marine animals and include animals as morphologically diverse as sea stars (Asteroidea), brittle stars (Ophiuroidea), sea cucumbers (Holothuroidea), sea urchins and sand dollars (Echinoidea), and sea lilies (Crinoidea). The name of the phylum is derived from the Greek "echinos," meaning spiny, and "derma," meaning skin. Echinoderms are deuterostomes and are therefore more closely related to chordates (which include ourselves) than they are to almost all other invertebrate phyla.
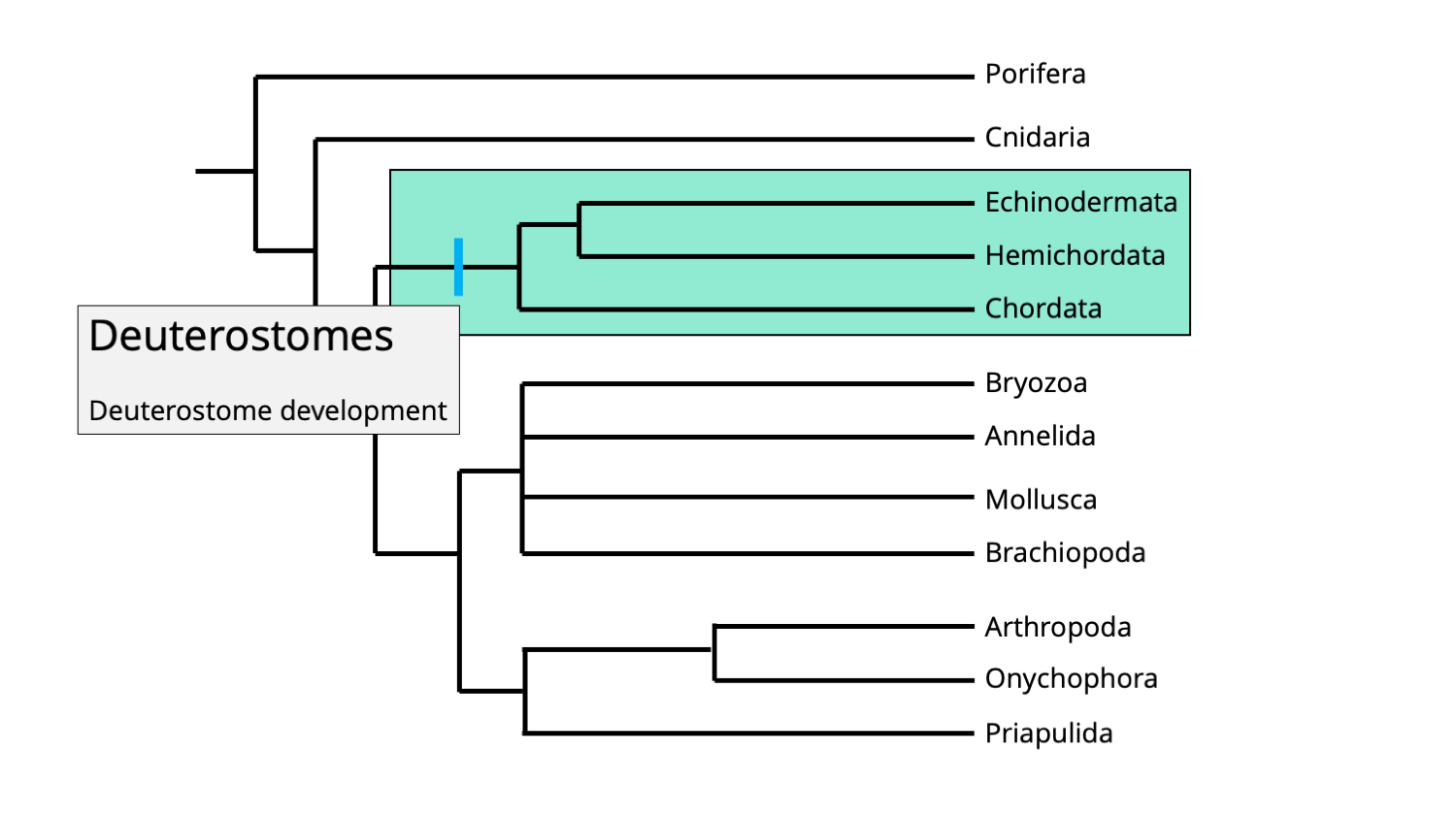
Phylogenetic positions of the bilaterian clade Deuterostomia, characterized by the development of the anus and then mouth during embryonic development. Image by Jonathan R. Hendricks, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
Adult echinoderms lack a sensory head region; however, they do have a nerve ring and possess various forms of sensory perception. Some groups have a simple one-way digestive system, but that hasn’t stopped them from occupying a great range of feeding modes. They are one of the most successful invertebrate groups and some classes (especially crinoids and echinoids) have substantial fossil records.
'A Success Story Echinoderms' by: Shape of Life (YouTube).
Phylogeny
Echinoderms are a diverse group with a variety of body forms, making it difficult to understand how the various classes are related. The earliest echinoderms arose in the Cambrian, with many more classes evolving by the end of the Ordovician, including all extant classes (learn more below in the section on fossil echinoderms).
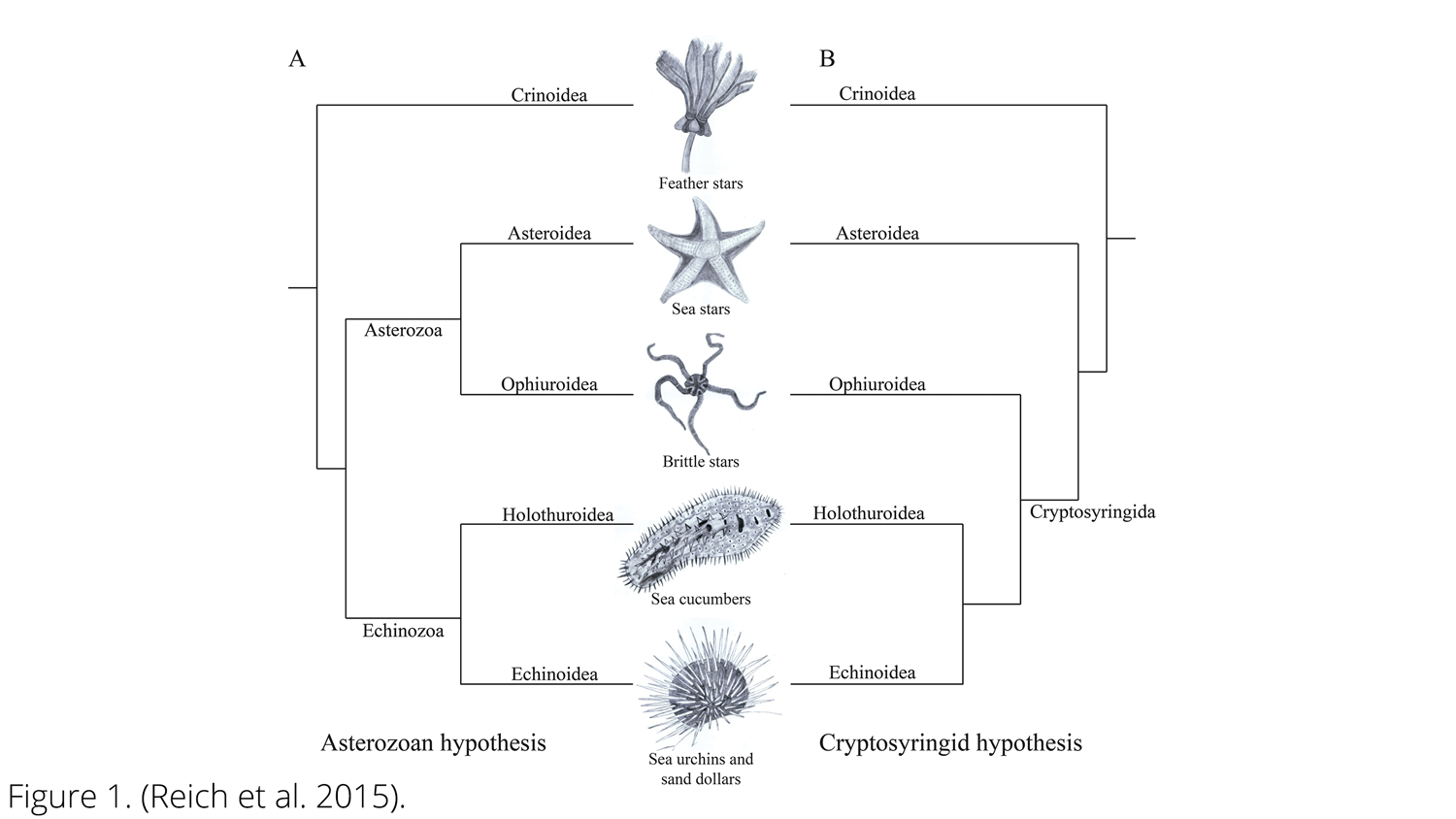
Image of competing echinoderm phylogenetic hypotheses. A: Asterozoan hypothesis B: Cryptosyringid hypothesis. (Reich et al., 2015; Creative Commons Attribution-ShareAlike 4.0 International License).
Several competing hypotheses to explain the phylogeny of echinoderms have been developed over the years, including two contrasting hypotheses for relationships among the five living classes. In one, called the Asterozoan hypothesis, Ophiuroidea and Asteroidea are sister taxa and form a clade called Asterozoa, whereas Holothuroidea and Echinoidea form a clade called Echinozoa (with Crinoidea sister to Asterozoa + Echinozoa). In the second, called the Cryptosyringid hypothesis, Ophiuroidea is placed as the sister taxon to Holothuroidea + Echinoidea. Recent molecular phylogenetic analyses (Reich et al., 2015; Telford et al., 2014) have supported the Asterozoan hypothesis, which we similarly favor here.
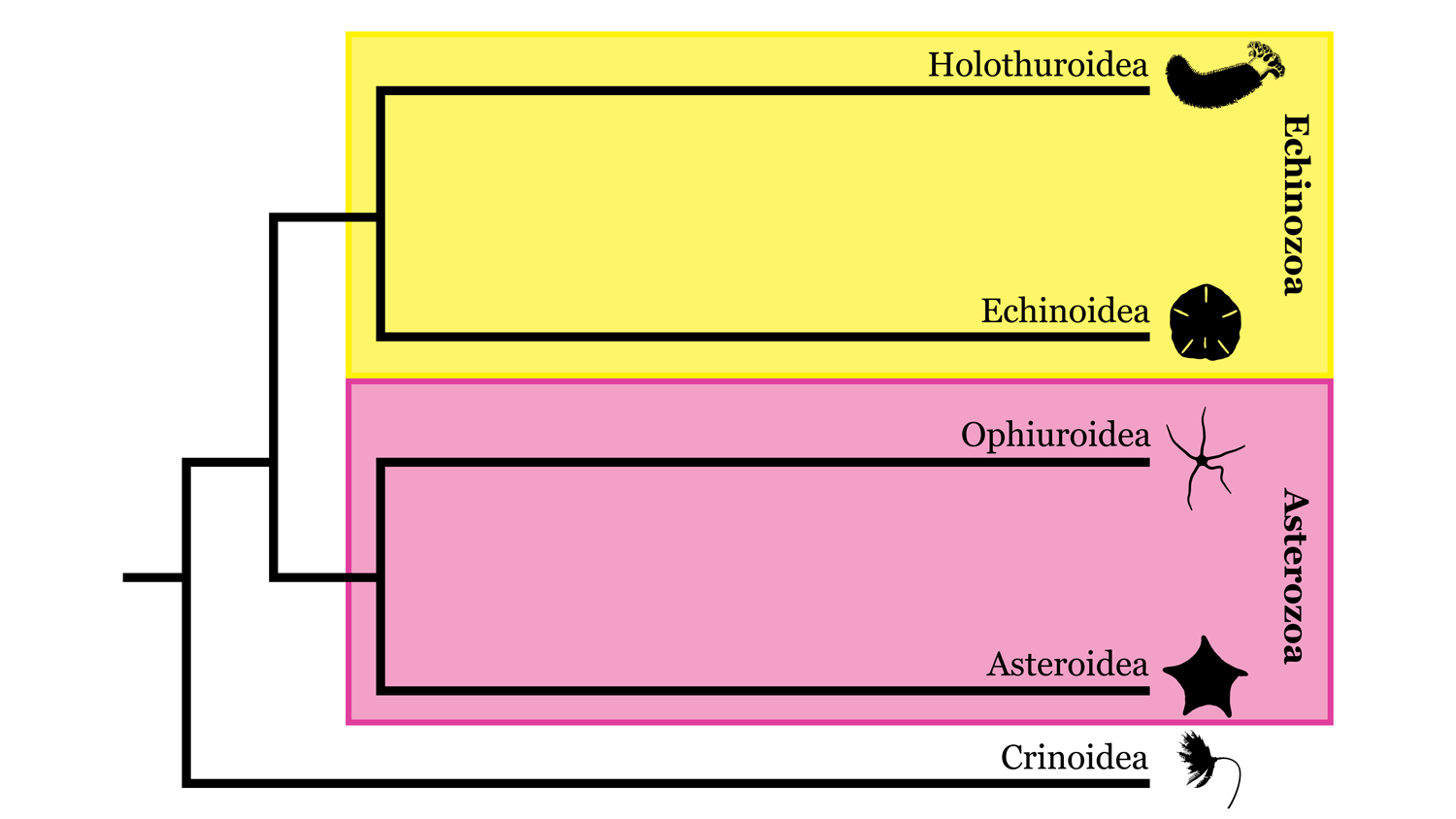
Highly simplified overview of Echinodermata phylogeny based in part on the hypothesis of relationships presented by Reich et al. (2015). Image by: Jaleigh Q. Pier, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
General Features
Pentaradial Symmetry
All adult forms of modern echinoderms have 5-point, or pentaradial symmetry, meaning that they have morphological features that are divisible by five. For example, starfish have five arms (or some multiple of five arms) and the calcareous endoskeletons (or, tests) of sea urchins can be similarly divided into fifths (see image below). Similarly, sand dollars have five "petals" on their dorsal side. What about the strange sea cucumbers? If you think of a sea cucumber as an elongated echinoid "test," they also possess pentaradial symmetry. It is important to note, however, that larval echinoderms often have bilateral symmetry (a synapomorphy for bilaterian animals; see here). Further, even though all extant classes of echinoderms have pentaradial symmetry, some extinct fossil forms from the early Paleozoic did not.
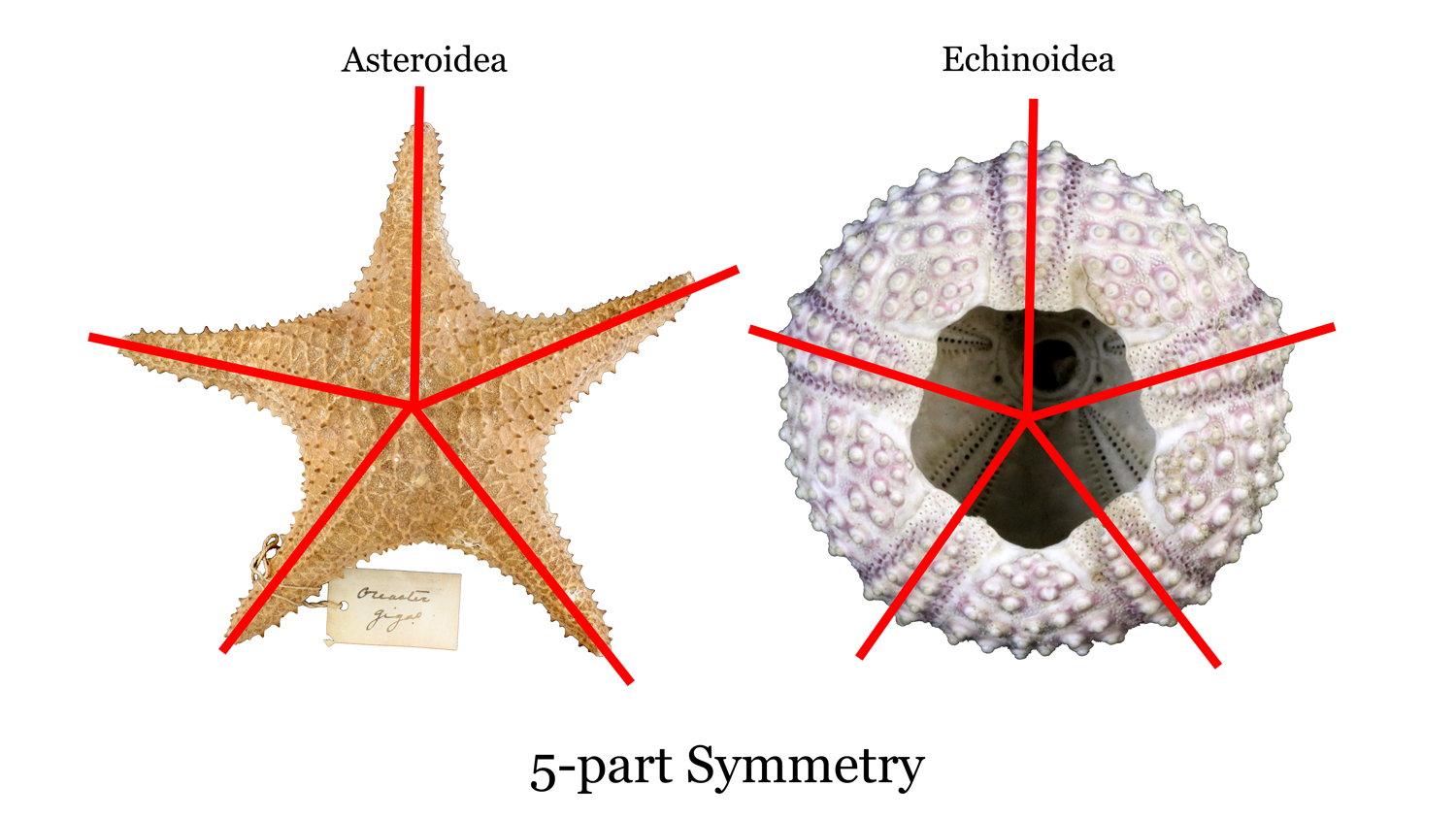
Image of pentaradial symmetry of both Asteroidea and Echinoidea. Image by: Jaleigh Q. Pier, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
If the concept of pentaradial symmetry seems difficult to grasp, watch the video below, which highlights the different 5-part symmetry of major echinoderm classes.
'Echinoderm Animation: Five-part symmetry' by: Shape of Life
Endoskeleton of ossicles and collagenous tissue
Echinoderms have a true endoskeleton made up of ossicles (i.e., small, rigid plates) of calcium carbonate with stereom microstructure that act as body support, sites of attachment for connective tissue, and protection. The dried-out skeleton is called a "test" (e.g., echinoids), "theca" (e.g., blastoids), or “calyx” (e.g., crinoids). Holothuroideans (sea cucumbers) are made up of primarily collagenous tissue and their ossicles are microscopic in size. Ossicles are connected by muscles and ligaments, which are made mostly of the protein collagen. Echinoderms have specialized mutable collagenous tissue, allowing them to change the rigidity of their skeleton. For example, they can become very hard and rigid to protect themselves from predators or soft and flexible when moving. Movement is achieved in various ways, including use of the tube feet of the water vascular system, ligament contractios for slow movements, and muscles contractions of rapid movement.

Image of echinoderm tests (Ophiuroidea, Asteroidea, Echinoidea) vs echinoderm theca (Blastoidea and Crinoidea). Image by: Jaleigh Q. Pier, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
Water Vascular System
The water vascular system is important for almost all life functions of modern echinoderms including movement, feeding, and respiration. Water pressure is controlled by this system to operate the tube feet, which are found in all extant echinoderm classes. These suction-cupped appendages are permeable to allow transfer of oxygen.
Typically, water pressure is controlled through the madreporite, which connects internally to the stone canal and then the ring canal. Numerous radial canals extend from the ring canal, often reflecting the 5-part symmetry of the echinoderm body plan. The tube feet extend from the radial canals and move via muscular contractions and relaxations of the ampullae.
Regeneration
A fascinating characteristic of all echinoderms is their ability to regenerate lost body parts. Both sea stars and brittle stars can regrow arms and, in some cases, divide in half to produce two new individuals. Some species of sea cucumber are also able to divide in half to create new individuals and can also regrow their entire digestive tract, which they can actively eject (more about this in the class Holothuroidea page!). Sea urchins are able to replace both spines and tube feet. Each of these regeneration methods will be discussed on the echinoderm class pages.

Photograph of a sea star regenerating at least seven arms (smaller, white-colored rays). Image by: Jonathan R. Hendricks, licensed under a Creative Commons Attribution-Share Alike 4.0 International License.
Fossil Record
Today, Echinodermata is a diverse phylum, with numerous species in five classes: Asteroidea (star fish), Crinoidea (sea lilies and feather stars), Echinoidea (sea urchins and sand dollars), Holothuroidea (sea cucumbers), and Ophiuroidea (brittle stars). In the fossil record, these classes are often represented by only pieces of their skeletons, which fall apart quickly after death because the various pieces are held together by soft tissues that decay. The pieces found include different components depending on the class:
- Crinoidea: disarticulated stem ossicles most commonly preserved, intact stem and arms preserved under exceptional conditions.
- Asteroidea: ossicle plates and endoskeleton commonly preserved, especially as molds.
- Ophiuroidea: central disk and ossicles most likely to be preserved intact, arms and body fossil preserved under exceptional conditions
- Echinoidea: loose spines and test most commonly preserved, body fossils under exceptional conditions.
- Holothuroidea: rarely preserved, however loose ossicles can be common.
The phylum has been represented exclusively by these five classes since the Triassic. Nonetheless, the fossil record of echinoderms includes as many as 28 additional classes, which originated and became extinct during the Paleozoic (see a few examples in the image below).
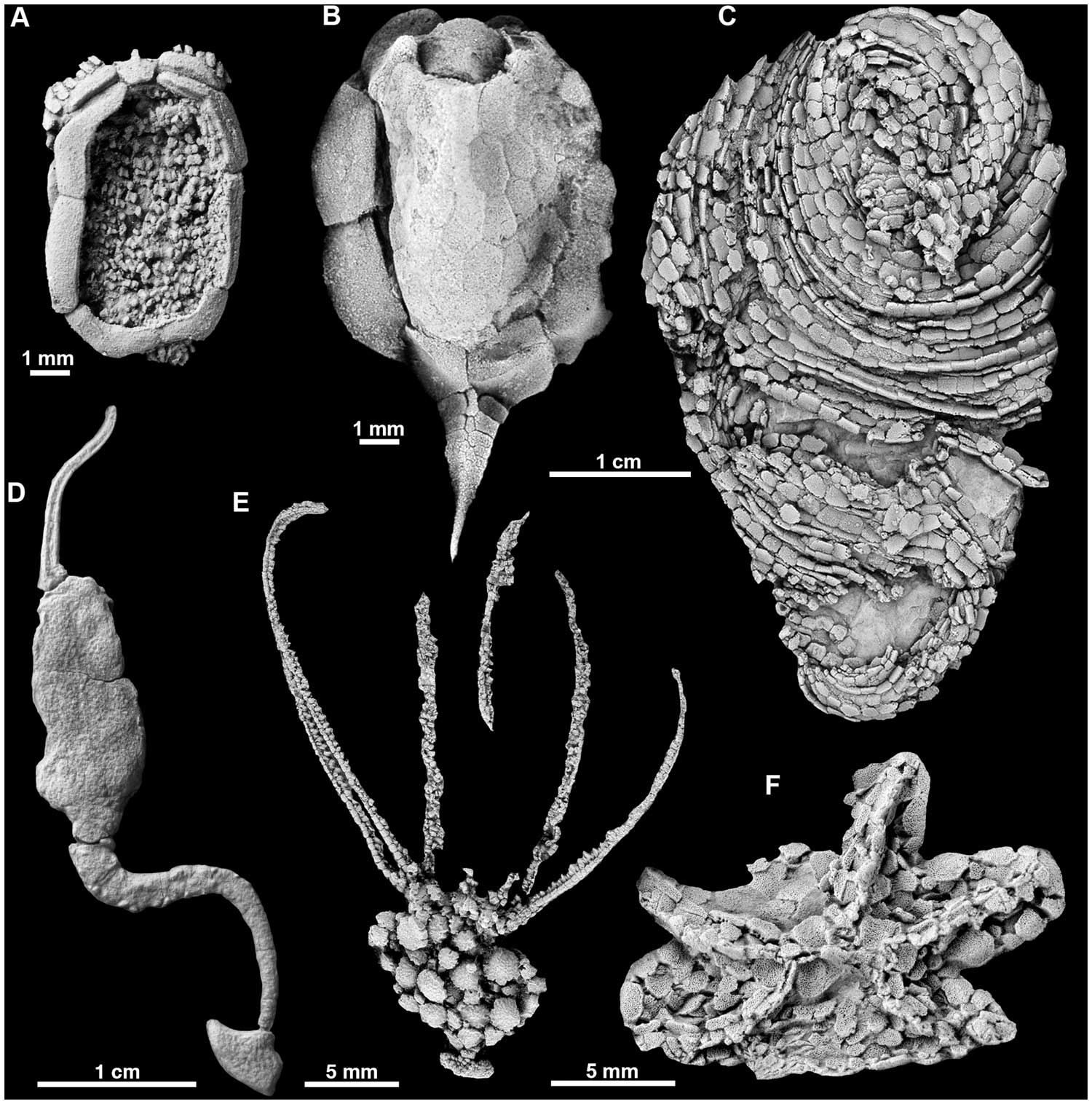
"Radiate and asymmetric echinoderms from the Cambrian showing a selection of primitive echinoderm body plans. A, the ctenocystoid Ctenocystis; B, the cinctan Gyrocystis; C, the helicoplacoid Helicoplacus; D, the solute Coleicarpus; E, the eocrinoid Gogia; F, stromatocystitid edrioasteroid." Figure and caption from Zamora et al. (2012) in PloS ONE; Creative Commons Attribution-Share Alike 4.0 International license.
The evolution of echinoderms can be thought of in four phases, as described by Ausich and Sumrall (2016) at the 9th European Conference on Echinoderms. The first phase of echinoderm evolutionary history begins with the evolution of echinoderm body plans during the Cambrian—putative claims of echinoderms from the Ediacaran (e.g., Arkarua) remain speculative, at best. As evidenced in the fossil record, early echinoderms exhibited a broad range of morphological diversity, including forms with helical, lateral, and pentaradial body plans. The second phase is marked by the diversification of species with all three body plans during the Cambrian, including both stalked and unstalked pentaradial forms. This diversification resulted in the first peak in class-level echinoderm diversity, with as many as 15 classes known from the later portion of the Cambrian.

Class-level diversity of echinoderms through time with important phases of echinoderm evolution indicated. Figure from Ausich and Sumrall (2016) presentation at the 9th European Conference on Echinoderms, reflecting the work of the Assembling the Echinoderm Tree of Life project. Included with permission from W. Ausich.
Several echinoderm classes were lost at the end of the Cambrian, but the class-level diversificiation during the third phase of echinoderm evolution resulted in as many as 21 classes during the Ordovician. It was during this time that the five extant classes first evolved. From the Late Ordovician to the end of the Permian, the number of echinoderm classes steadily decreased. In addition to the numerous extinct Paleozoic classes, the end-Permian mass extinction nearly wiped out the crinoids and echinoids, with some evidence suggesting as few as one family from each class survived this catastrophic event. The fourth phase of echinoderm evolution began in the Triassic and is defined the diversification within the five remaining, extant classes.
As briefly introduced here, not all of the diverse echinoderm morphologies evolved at the same time, suggesting that the defining features of this phylum, and potentially other phyla, were not set in stone, so to speak, at the first appearance in the fossil record. Instead, different classes arose over millions of years, with differential extinction ultimately filtering out those groups we might consider unusual, and scientifically fascinating, today (see the image above). Because of their limited fossil record, the phylogenetic relationships between these groups remain challenging to piece together (see one hypothesis in the image below). Understanding these relationships has been the focus of a large, collaborative research project called Assembling the Echinoderm Tree of Life—visit their webpage to see what progress has been made recently!
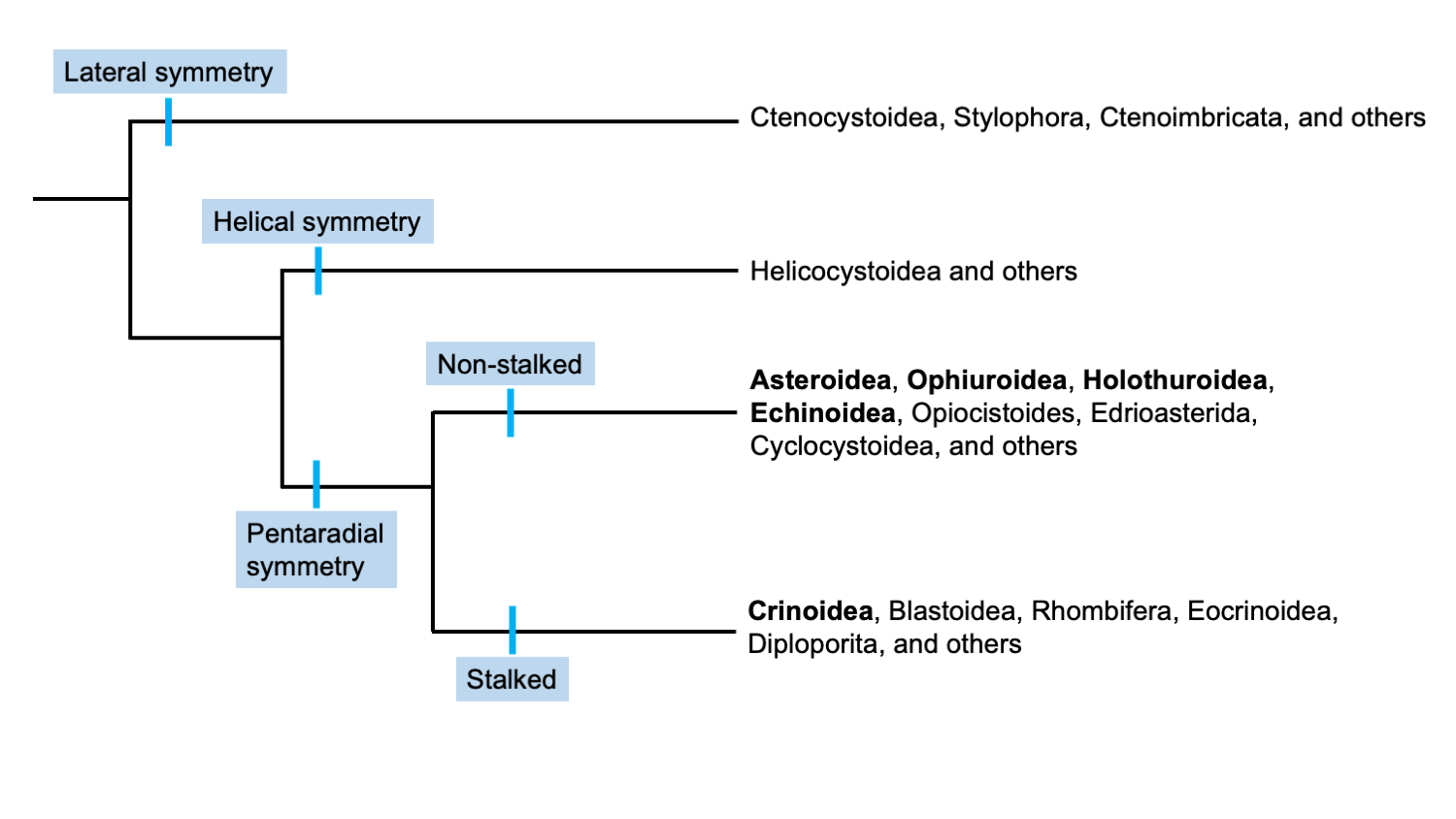
A hypothesis on the relationship between different groups of echinoderms based on symmetry. Note, living classes are in bold. Figure modified from Ausich and Sumrall (2016) presentation at the 9th European Conference on Echinoderms, reflecting the work of the Assembling the Echinoderm Tree of Life project. Included with permission from W. Ausich.
In assessing the evolution and development of this phylum, Deline et al. (2020) identified four general echinoderm morphologies: (1) non-radial; (2) stalked radial crinoids; (3) stalked radial blastozoans; and, (4) pentaradial directly attached and mobile. For the purposes of this chapter, we will consider the second and third groups together (i.e., stalked radial), as they are highly similar in appearance and likely form a clade (i.e., Pelmatozoa). In the pages that follow, we will take a closer look at the extant classes and, first, a selection of exclusively fossil classes within these three morphological groups: non-radial, stalked radial (crinoids and blastozoans), and pentaradial attached and mobile.
Acknowledgments
We thank Bill Ausich, Jen Bauer, and Sarah Sheffield for providing feedback on earlier versions of this chapter to improve its quality.
References and further reading:
Boardman, R. S., A. H. Cheetham, and A. J. Rowell. 1987. Fossil Invertebrates. Blackwell Scientific Publications. 713 pp.
Deline, B., J. R. Thompson, N. S. Smith, S. Zamora, I. A. Rahman, S. L. Sheffield, W. I. Ausich, T. W. Kammer, and C. D. Sumrall. 2020. Evolution and Development at the Origin of a Phylum. Current Biology, 30: 1672-1679.
Nichols, D., 1967. Echinoderms. Hutchinson University Library, London.
Reich, A., C. Dunn, K. Akasaka, G. Wessel. 2015. Phylogenomic Analyses of Echinodermata Support the Sister Groups of Asterozoa and Echinozoa. PLoSONE, 10: e0119627.
Smith, A. B., S. Zamora, and J. J. Alvaro. 2013. The oldest echinoderm faunas from Gondwana show that echinoderm body plan diversification was rapid. Nature Communications, 4: 1-7.
Telford, M. J., C. J. Lowe, C. B. Cameron, O. Ortega-Martinez, J. Aronowicz, P. Oliveri, and R. R. Copley. 2014. Phylogenomic analysis of echinoderm class relationships supports Asterozoa. Proceedings of the Royal Society B: Biological Sciences, 281: 20140479.
Wright, D. F., W. I. Ausich, S. R. Cole, M. E. Peter, and E. C. Rhenberg. 2017. Phylogenetic taxonomy and classification of the Crinoidea (Echinodermata). Journal of Paleontology, 91: 829-846.
Zamora, S., B. Deline, J. J. Álvaro, and I. A. Rahman. 2017. The Cambrian Substrate Revolution and the early evolution of attachment in suspension-feeding echinoderms. Earth-Science Reviews, 171: 478-491.
Usage
Unless otherwise indicated, the written and visual content on this page is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. This page was written by Jaleigh Q. Pier and Jansen Smith. See captions of individual images for attributions. See original source material for licenses associated with video and/or 3D model content.




