Chapter contents:
Evolution and the Fossil Record
– 1. Natural selection ←
– 2. Species and species concepts
– 3. Speciation
– 4. Punctuated equilibria and stasis
–– 4.1 Videos about punctuated equilibrium and stasis
– 5. Macroevolution
–– 5.1 Hierarchies
–– 5.2 Species selection
–– 5.3 Abiotic vs. biotic causes of macroevolution
–– 5.4 Evolutionary radiations
Above image: A marble statue of Charles Darwin on display at the Natural History Museum, London and Darwin's famous sketch of an evolutionary tree, labeled "I think."
Charles Darwin
Numerous achievements make Charles Darwin (1809–1882) a giant in the history of science, every part the equal of Galileo Galilei, Isaac Newton, and Albert Einstein. First, he was a prominent, well-respected, and well-connected scientist who was active during a time when—at least among scientists—there was a groundswell of support growing for evolution. Darwin was also very wealthy. He was born to a rich family and married a rich woman (Emma Wedgwood). Because of this, he did not have to work for a living, allowing him a luxury of time and resources to investigate and think about the natural world. Second, Darwin's On the Origin of Species provided in a single English-language volume an overwhelming catalog of evidence in support of the fact of evolution. Darwin was not the first person to "connect the dots" and discover that evolution happened, but he did put forth a prodigious amount of material evidence in support of those dots and explain how they were connected. Third, Darwin discovered—or co-discovered, depending on whom you ask—the theory of natural selection, the mechanism responsible for explaining the goodness of fit (or, adaptation) of organisms to the environments in which they live.
Darwin is among the most carefully studied figures in the history of science. Numerous biographies have explored his life and his own personal evolution as a naturalist and scientist. Indeed, nearly every letter, note, and scribble produced by Darwin has been investigated and most of these can be explored via the Darwin Manuscript Project at the American Museum of Natural History.
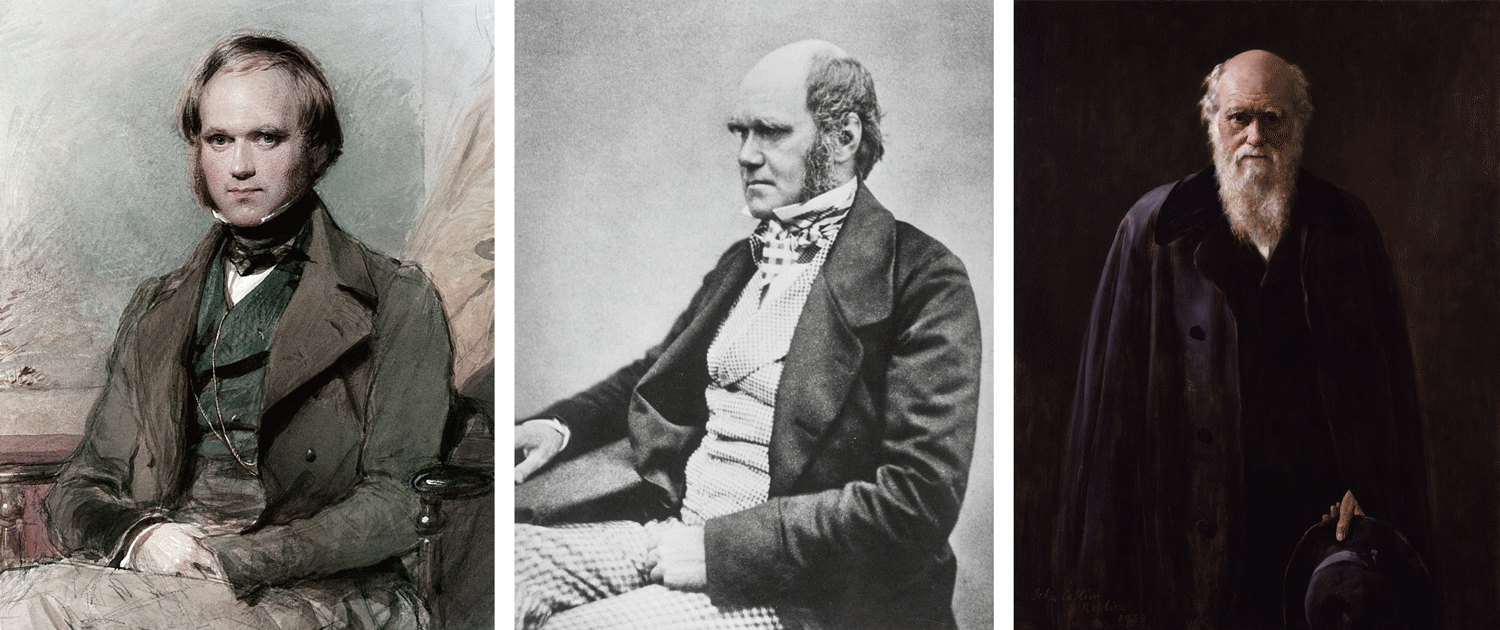
Charles Darwin during different stages of his life. Left: portrait of Darwin as a young man in the late 1830's (by George Richmond). Middle: photograph of Darwin in middle age (likely 1854). Right: portrait of Darwin in old age (by John Collier; 1881). All images from Wikipedia/Wikimedia Commons (public domain).
We are not going to cover Darwin's fascinating personal story in detail here, as there are many other excellent resources available elsewhere that explore his early life, the famous Voyage of the Beagle that exposed Darwin to the grandeur of the natural world as a 20-something young man, and the many years that he spent afterwards agonizing about making public his case for evolution and natural selection (see "References and further reading below). In particular, we recommend the short, engaging account of Darwin's life by Sean Carroll in the book "Remarkable Creatures: Epic Adventures in the Search for the Origin of Species." We also recommend the lecture "Darwin's Revolution in Thought" by Stephen Jay Gould. The quality of the video is not good, but this is one of the finest introductions available to Darwin's life, how natural selection works, and why the idea of natural selection is so revolutionary.
"Darwin's Revolution in Thought" by Stephen Jay Gould. Source: Youtube.
Here, we wish to focus upon Darwin's most important discovery: natural selection itself. Before we do, though, we want to first mention a somewhat under-appreciated scientist, Alfred Russel Wallace, who also deserves co-credit for its discovery.
Alfred Wallace
Alfred Russel Wallace (1823–1913) was a contemporary of Charles Darwin (though 14 years younger) and an accomplished naturalist in his own right. From the very beginning, Wallace has always been recognized as a co-discoverer of natural selection. But Wallace was poor and little known when he published his ideas on evolution, while Charles Darwin was rich and already famous when he did the same.
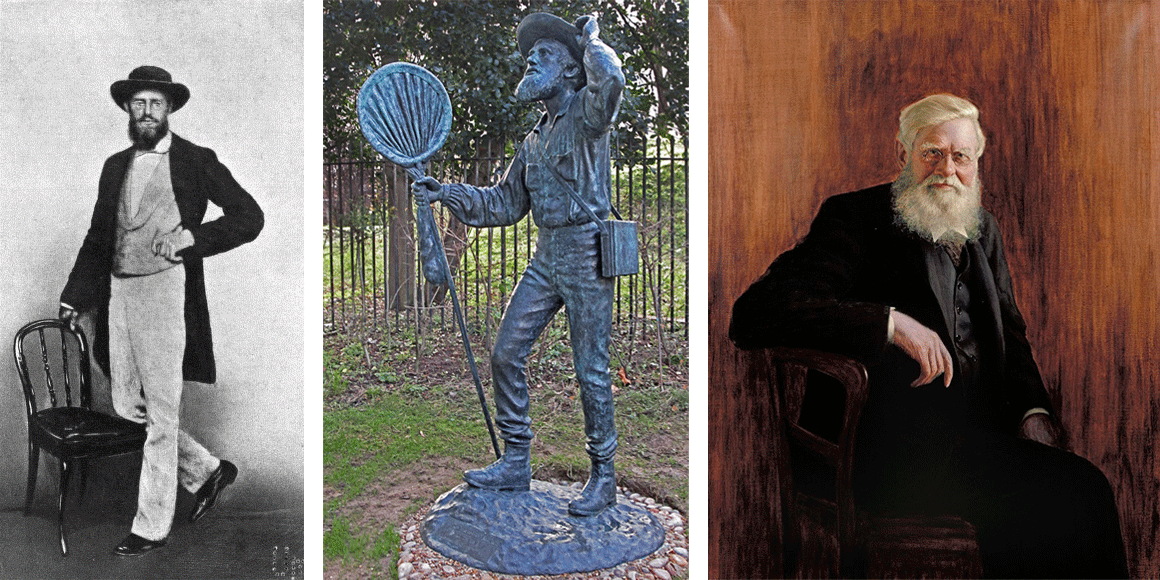
Alfred Wallace during different stages of his life. Left: photograph of Wallace during middle age, taken in Singapore in 1862. Middle: bronze statue of Wallace, showing him as a field naturalist (ca. 1859); statue created by Anthony Smith. Right: portrait of Wallace as an older man (by John William Beaufort). Left and right images from Wikipedia/Wikimedia Commons (public domain). Middle photograph by George Beccaloni; Creative Commons Attribution-Share Alike 3.0 Unported license.
This difference in fame and connections partly explains why Wallace is often treated as a footnote in discussions of evolution, while Darwin is presented front and center. Be that as may, we feel it is important to discuss how Wallace published cogent evidence in 1855 that evolution had happened—four years before Darwin did (On the Origin of Species was published in 1859)—in the article “On the Law which has Regulated the Introduction of New Species” in the Annals of Natural History. You may access this important paper in its original formatting on the Biological Heritage Library or on a website developed by Charles Smith at Western Kentucky University (his broader Alfred Russel Wallace Page contains all the publications of Wallace, along with exceptional commentary, and a plethora of additional information about Wallace’s life and writings).
Wallace’s 1855 work described evidence from modern and fossil organisms that evolution had happened. Before 1855, Darwin had been content to share his musings on evolution with only his closest friends (including Charles Lyell), in spite of the fact that he had been jotting down ideas about the subject in private diaries since 1837.

Darwin's famous 1837 notebook sketch of an evolutionary tree, labeled "I think." The interpreted handwriting associated with the sketch reads, "Case must be that one generation should have as many living as now. To do this and to have as many species in same genus (as is) requires extinction. Thus between A + B the immense gap of relation. C + B the finest gradation. B+D rather greater distinction. Thus genera would be formed. Bearing relation [next page begins] to ancient types with several extinct forms." Source: Wikipedia/Wikimedia Commons (public domain). Also see the Darwin Online webpage.
Darwin saw Wallace’s (1855) paper and several of his friends urged him to publish on his views, but for some reason he did not. This changed in 1858 when Wallace, ensconced half way around the world on Ternate Island (in what is today Indonesia), mailed to Charles Darwin, of all people, a manuscript he had written that he hoped Darwin would help present and publish on his behalf. This is especially surprising because Wallace had no idea the extent to which Darwin had already thought about and developed his ideas on evolution.
Google map showing the location of Ternate Island, Indonesia.
The manuscript that Wallace sent to Darwin (received June 18, 1858) detailed a mechanism for evolutionary change that was eerily similar to Darwin’s own theory of natural selection. While Wallace was incommunicado in Indonesia, Darwin had colleagues Sir Charles Lyell (of uniformitarianism fame) and botanist Joseph Hooker present Wallace's manuscript at a July 1, 1858 scientific meeting of the Linnean Society of London.
Wallace's manuscript was not, however, presented unchanged. Darwin added himself as first author to the manuscript (see image below) and also added a large amount of text detailing his own ideas on natural selection taken from what would become his Origin of Species manuscript, as well as passages from a letter that he had sent to American botanist Asa Gray. The latter—as well as the introductory affirmations of Lyell and Hooker—were presented as evidence that Darwin had already been working for years on the topics of evolution and natural selection, long before Wallace began thinking about the subjects. This classic piece of scientific political maneuvering may be explored from the original on the Biodiversity Heritage Library.

The first two pages of "On the tendency of species to form Varieties; and on the Perpetuation of varieties and species by natural means of selection" by Charles Darwin and Alfred Wallace (July 1, 1858). Source: Biodiversity Heritage Library (public domain).
Ironically, the president of the Linnean Society remarked in his May 1859 Presidential address that:
"The year which has passed [1858]… has not, indeed, been marked by any of those striking discoveries which at once revolutionise, so to speak, the department of science on which they bear." - Thomas Bell.
That statement surely can be consigned to the dustbin of history, as 1858 represents the first publication on natural selection, and further, Wallace’s letter and the manuscript it contained ended up being the impetus for Darwin to publish On the Origin of Species in 1859. This singular event forever impacted human views about our place in nature. But we believe that Wallace deserves greater recognition for contributing to this scientific revolution than he has received.
Wallace did subsequently become well known, in part due to a book he published in 1869, “The Malay Archipelago,” that described his experiences as a field naturalist in that region.

The title page of Wallace's 1869 book "The Malay Archipelago," along with two images from inside the book, the skull of the deer-pig (Babirusa) and Wallace's flying frog (Rhacophorus nigropalmatus; see video below about this frog). All images from Wikipedia/Wikimedia Commons (public domain).
Wallace's Flying Frog - Bill Bailey's Jungle Hero - Episode 1. Source: BBC Two (YouTube).
Wallace and Darwin became lifelong friends and Wallace seemingly did not care who got the majority of the credit for the idea of natural selection. He simply wanted the idea to get out in the scientific literature and thereby be considered as a mechanism for evolutionary change. In many respects, Wallace seems an amazing embodiment of what science should be about.
Natural selection
The co-discovery of natural selection by Darwin and Wallace is one of the great achievements of scientific study. The idea is also very simple and intuitive, nearly to the point of being obvious once clearly articulated. Indeed, upon learning about natural selection, zoologist T.H. Huxley remarked, "How extremely stupid of me not to have thought of that."
Let's now consider natural selection—which can be thought of as a statistical law—in all of its simplicity. The idea has four basic principles.
1. Each generation, more organisms are born than survive.
This situation arises because members of the same species compete for similar resources and these resources—especially food—are limited in nature.
Let's consider the case of the humble—albeit not particularly beloved—German cockroach, which is a common pest species.

The German cockroach Blattella germanica. Photograph by "Lmbuga" (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
"Cockroach Infestation" by National Geographic (YouTube).
Fertilized female German cockroaches carry a structure inside their bodies called an ootheca that contains ~50 developing eggs (Source: Orkin.com); after hatching and going through numerous molts, these develop into adult cockroaches in about 50-60 days (Wikipedia). Under a model of exponential population growth and unlimited food resources, cockroaches would soon seemingly take over the world if each individual that was born survived to adulthood and also reproduced. Luckily for us, the resources available for cockroaches are limited and many that are born do not survive to adulthood.
2. Organisms vary
No two organisms are exactly alike. If you compare members of any species, you will notice subtle differences between individuals. This fact is obvious to us, as all humans have slightly different appearances (we vary in height, weight, eye color, hair color, and numerous other factors). Individual cockroaches also vary relative to one another. Some are slightly larger, faster, or heavier than others. They may also vary in their food preferences (for example, some might—at least according to urban legend—have a particular taste for your eyelashes or fingernails.).

The cave-dwelling orange-headed cockroach Eublaberus posticus, which feed on dead bats (and sometimes the elytra of their companions). On exhibit at the Museum of the Earth, Ithaca, New York. Photograph by Jonathan R. Hendricks.
Carefully observe the plants and animals that live in your neighborhood. Once you start looking, you will observe that variation within species is a rule of nature. Ultimately, this natural variation is caused by genetic mutations (or, errors in the genetic code).
3. Some variation is heritable
Some, but not all, of the differences that characterize individual organisms may be passed on to the next generation (such traits are said to be heritable). For example, fast cockroaches might have offspring that are also fast, if speed is a heritable trait.
Think about your own body features and which are similar to those of your mother or father. When we say things like "she has her mother's eyes" or "he has his father's nose," we are making statements about heritability and traits that run in families. The same thing occurs in nature.
4. Organisms that survive and reproduce often have the most favorable variations
Organisms that have heritable variations that offer even the slightest survival advantage or reproductive advantage in the environments in which they live are most likely to contribute their advantageous genes to future generations.
In the case of cockroaches, being slightly faster might allow individuals to better procure food relative to their slower compatriots, or to escape a shoe headed down towards them. Others might be slightly more tolerant of cold temperatures, favoring their survival in frigid new habitats (consider this news story from New York regarding a newly-introduced species of cockroach that is adept at surviving freezing temperatures).
"Cockroaches Survive Squeezing, Smashing, and More" by National Geographic (YouTube).
To summarize, natural selection states that because natural resources (such as food) are limited, organisms with even subtly advantageous, heritable variations are more likely to survive and reproduce relative to individuals that do not have those variations. In other words, organisms with favorable traits survive and contribute those traits to the next generation, making the traits more common in a population over time. This natural process has also been aptly characterized as "survival of the fittest" (often attributed to Darwin, but coined by Herbert Spencer).
Some opponents of evolution have characterized the process as being completely random, arguing therefore that it would be impossible for anything complex to evolve through incremental random steps. As you have learned, however, natural selection is anything but random; in fact, it is just the opposite. Further, evolution operates in steps, building upon what came before, but never starting over from scratch.
Many complex questions extend from the constant operation of natural selection. For instance, is it competition among organisms that poses the strongest selective force, or is it the effects of the physical environment? Organisms can be selected (they survive and reproduce, or die), but can things like genes or species be selected? What typically happens to species morphology through time in the face of such selection? We will take up all of these topics later in this chapter.
Artificial selection
Darwin began On the Origin of Species with a chapter titled, "Variation under Domestication." He knew that his ideas surrounding evolution would be controversial for most of his readers, so he began with a subject that would be approachable and understandable for most of his audience.
People with knowledge of animal husbandry and agriculture were well aware that different varieties of domesticated animals and crop plants with particular beneficial or appealing features had been selectively bred from wild ancestors, enhancing those features over time. For example, a farmer might select the cow that produced the most milk to be the mother of the next generation of calves, thereby increasing the milk production of their herd over time. We have come to know this process as artificial selection and it is analogous to natural selection; the only difference is the importance of human action in artificial selection, and its absence in natural selection.
In the first chapter of the Origin, Darwin goes on at great length about all of the varieties of pigeons which had been generated through the action of artificial selection by breeders. Keeping and breeding pigeons was all the rage in Victorian England and continues to be an enjoyable hobby for some today.

A pigeon in Basel, Switzerland. Photograph by Jonathan R. Hendricks.
"See How Pigeons Saved This Man From a Life on the Streets | Short Film Showcase" by National Geographic (YouTube).
Dogs provide excellent evidence of the power of artificial selection. Consider the many shapes and forms of different dog breeds, all produced by artificial selection for prized traits. Some of these traits were selected for hunting and guarding, while others were developed seemingly just to amuse us.

A small sampling of the diversity of modern dog breeds. Image from Shearin and Ostrander (2010) in PLoS Biology. Creative Commons Attribution-Share Alike 4.0 International license.

Left: Variation in the adult sizes of two dog breeds. Right: coat variations in four dog breeds. Images from Shearin and Ostrander (2010) in PLoS Biology. Creative Commons Attribution-Share Alike 4.0 International license.
Based on genetic comparisons, scientists are nearly certain that dogs were domesticated from wolves; this likely happened around 15,000 years ago, and perhaps earlier. The number of different dog breeds, however, is a relatively recent phenomenon, with most originating in the past 200 years, largely as a result of cross breeding several ancestral lineages of domesticated dog.
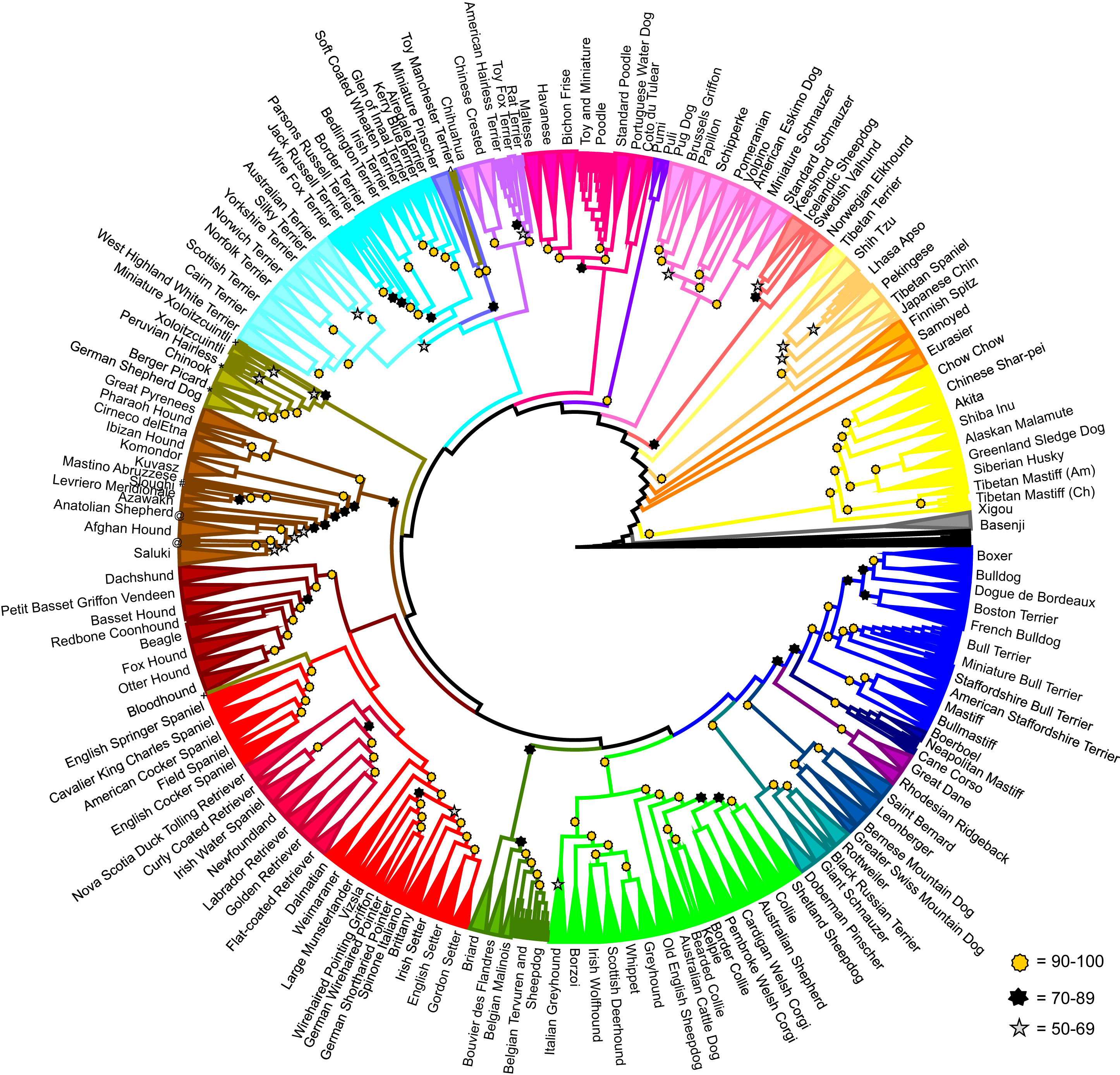
Relationships of 161 domestic dog breeds based on analysis of genetics; examples of some of these breeds are shown below. Image from Parker et al. (2017) in Cell Reports. Creative Commons Attribution License (CC BY).

Some of the dog breeds analyzed by Parker et al. (2017) (see corresponding color-coded clades above). Image from Parker et al. (2017) in Cell Reports. Creative Commons Attribution License (CC BY).
The importance of Darwin's argument with respect to artificial selection is that if humans could produce such extensive changes to animals and plants over the course of a few decades to centuries, then incredible amounts of change could be produced by nature over the immensity of geological time.
Adaptations, exaptations, and spandrels
Adaptations
An adaptation is a feature of an organism—resulting from natural selection—that allows them to play a particular function or perform a task that confers a survival or reproductive advantage relative to organisms that lack the feature. Adaptations must be features that were selected for that function, both in present day populations, as well as in the ancestral populations within which the feature first evolved.
We can call the vertebrate eye an adaptation for complex vision because we can easily understand how it enabled vertebrates to make much more detailed assessments of the surrounding environment relative to the eyes of their ancient ancestors that could detect light, but not form distinct images.

Eye of a golden eagle (Aquila chrysaetos). Photograph by Peter Kaminski (Wikipedia/Wikimedia Commons; Creative Commons Attribution 2.0 Generic license).
It is entirely plausible to think that the acquisition of complex eyes and the ability to see more complete images would provide a selective advantage for organisms that possessed this trait relative to those that did not. Vision makes it easier to find food and escape predators, and thus survive and eventually reproduce.
Among most modern vertebrates, any reduction, or loss, of the ability to see will generally make it harder to obtain food or escape predators. (Thankfully glasses exist, or the authors of this piece would be in serious trouble!) Thus, there is active selection pressure that maintains the effective functioning of vertebrate eyes in modern populations. This point can be driven home by the fact that vertebrates can be found in a few environments where light is completely absent (typically in caves or deep underground) and vision is not useful. Some vertebrates in such environments have lost functional eyes. An interesting example is the freshwater Mexican tetra fish, Astyanax mexicanus. Populations of this species that live in sun-lit surface waters have typical fish eyes and corresponding visual capabilities. Multiple populations of this same species have adapted to living in dark cave environments, however, and have independently gone completely blind.

The Mexican tetra, Astyanax mexicanus. Left: surface forms of the fish, which have typical eyes; image by Clinton and Charles Robertson (Wikipedia/Wikimedia Commons; Creative Commons Attribution 2.0 Generic license). Right: cave form of the fish, which do not have eyes; image by H. Zell (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
The loss of vision in cave-dwelling species likely happens for one of two reasons. The first is that there might be some physiological cost to having eyes, such that organisms that no longer have to gather the food and energy necessary to maintain their eyes (finding and consuming all those carrots … ) may be better able to survive or reproduce. The second is that any mutations that cause changes in the ability to see may no longer affect an organism’s ability to survive or reproduce; thus, the mutations are selectively neutral such that they will proliferate within a population effectively for chance reasons. The latter is related to the fascinating idea of "neutral evolution," which focuses on changes at the molecular level and was developed by Motoo Kimura. Although we will not discuss this idea in detail here, we will discuss the related role of chance and random factors at the larger macroevolutionary scale in the next chapter on extinction.
Opponents of evolution have long wondered how complex structures like eyes could have arisen by chance (see Talk Origins). The answer, of course, is that the complex eyes of humans and eagles did not arise all at once as a variation in an otherwise blind population of some ancestral vertebrate. Instead, the vertebrate eye arose through a series of steps, each of which occurred a very long time ago.
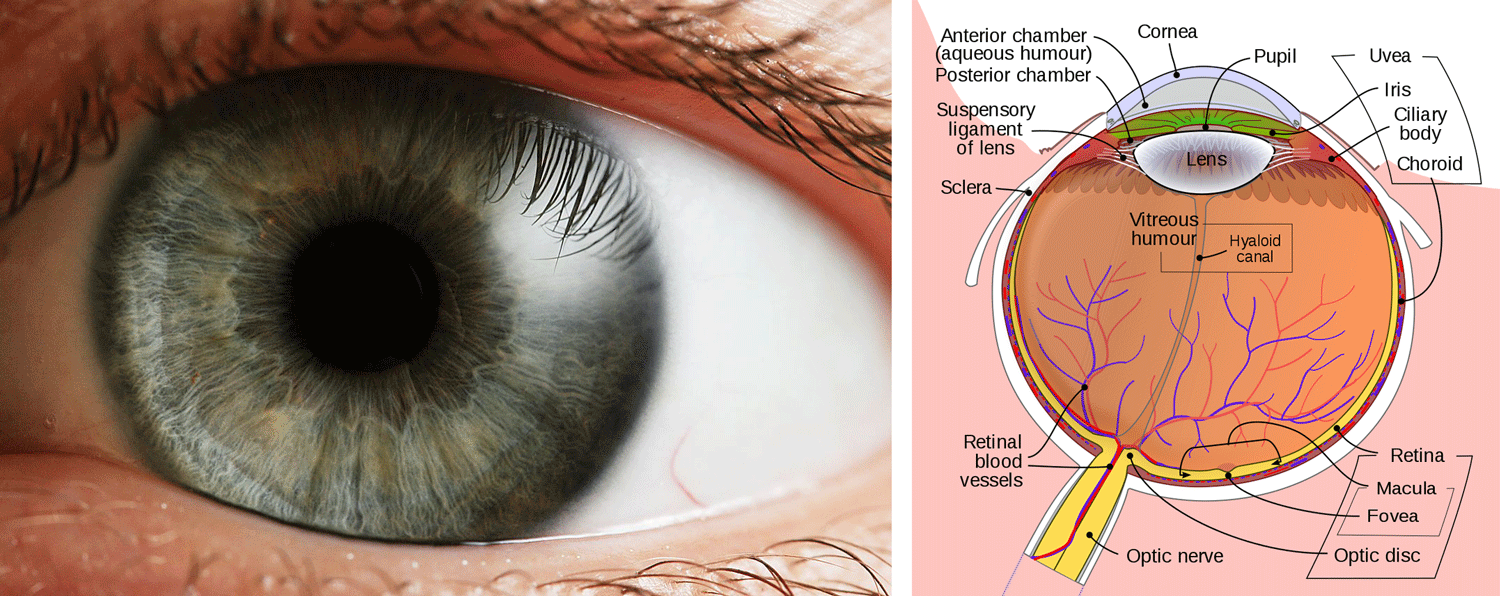
Left: photograph of a human eye, showing details of the iris surrounding the pupil; image by "che" (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 2.5 Generic license). Right: parts of the human eye, as seen from above; image by "Rhcastihos" and "Jmarchn" (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
Several parts of the eye are individual adaptations. The iris is that part of the eye that is colored; when we say that a person has blue eyes, what we mean is that they have blue irises. The function of the iris is to filter the amount of light that reaches the retina at the back of the eye, thereby maintaining the acuity of vision in differing light levels. (If you have ever had your eyes dilated during an eye exam, you know how difficult it is to see outside on a sunny day when your pupils are wide open.) We could share videos of human eyes dilating, but this is probably more enjoyable to study in playful cats.
"cat eyes dilation before striking" by c2chung (YouTube).
The individual parts of the eye, and the eye as a whole, are still being shaped by selection today. Consider that an organism that had eyes with an iris that functioned less effectively might be slightly less adept at surviving or reproducing. Over the long term, we would expect better-functioning irises to become dominant in a population relative to those that function less well.
There are also examples of adaptations that evolved on much more recent time scales than the vertebrate eye. Indeed, an adaptation doesn’t have to be something extremely complicated that evolved a long time ago. For example, there are sites polluted by humans within the last 100 years that are contaminated with toxic compounds, such as heavy metals or oil. Some microbes in these environments have very recently evolved the ability to break down these compounds, which would otherwise be fatal to most life forms. In fact, these microbes are now sometimes used to clean up such contaminated sites (this is called bioremediation). Some mutation or series of mutations happened in the chemical pathway of these bacteria that allow them to digest otherwise toxic compounds. The evolution of this pathway made them better able to survive and reproduce, such that they proliferated in environments that exclude many other potential competitors.
Exaptations
While this discussion of adaptations might seem straight forward, it is common to encounter the term being used loosely as an assumption, rather than as a statement supported by evidence or testing. For example, someone might say, "feature X evolved to do Y." The problem with such statements is that there is usually no consideration of potential selection pressure, the timing of when "feature X" evolved, or even whether the feature actually functions in the manner posited.
For example, consider the statement, "eyebrows in humans evolved to keep sweat out of our eyes." If so, then: 1) Were there ancient humans who lacked eye brows and therefore had a survival and reproductive disadvantage because they got so much sweat in their eyes?; 2) What if (and this is probably not the case) sweating evolved after eyebrows did?; and 3) Do eyebrows actually help to keep sweat out of the eyes? (This is very testable, but some people are going to have to shave their eyebrows off and then run a lot of laps to figure out the answer.)
In a general sense, it may be fine to make these types of statements as a matter of informal shorthand (e.g., in a public setting), with the understanding that of course a feature doesn’t evolve to do something (instead, there may be selection to maintain a newly evolved variation within a population). It is important to understand, however, that the term adaptation has a precise meaning in a scientific context (see above). Further, for a very long time following the publication of On the Origin of Species, it was assumed that every feature of an organism must have been crafted as an adaptation for some purpose that favored survival and/or reproduction. This mindset has specific implications about the patterns of evolution we would thereby predict and our understanding of the nature of evolution. However, the proposition that all traits are adaptations was often not tested when it came to the presumed selective advantage associated with the trait, nor was the historical context of the trait considered.
Let's explore an example of purported adaptation that has captivated biologists and paleontologists for many years: "feathers evolved in birds so that they could fly" (or, worded another way, "feathers are an adaptation for flight").

A great horned owl (Bubo virginianus), using its feathers for flight. Photograph by Peter K. Burian (Wikipedia/Wikimedia Commons; Creative Commons Attribution 4.0 International license).
Feathers are indeed critical for flight in all living bird species that take to the air (some birds like penguins and ostriches, of course, do not fly). They also provide insolation for warmth (there is a reason why people in cold climates wear goose-down jackets and put down comforters on their beds) and are sometimes brightly colored or distinctively patterned to help individuals recognize members of their own species. But, did feathers first evolve for flight? That is, are they an adaptation for flight? Studies of dinosaur fossils help to answer this question.
Consider Tyrannosaurus rex, the most famous and iconic dinosaur, which lived in western North America during the very end of the Cretaceous period, about 66 million years ago.

Tyrannosaurus rex, on display at the American Museum of Natural History in New York City. Photograph by Jonathan R. Hendricks.
This fearsome animal may have weighed up to 8 tons but had famously small arms (about the same length as yours).
"Dinosaur Scene - big head little arms" from the movie Meet the Robinsons (YouTube).
Clearly, T. rex was not an animal that could fly: it is just not structurally possible. Yet, surprisingly, we know now that Yutyrannus huali, a close cousin of T. rex, had a body covered in feathers. It is very likely that T. rex did as well, especially early in its life (see recent reconstructions here, as well as the video below).
"What Did a Baby T. rex Look Like?" by American Museum of Natural History (YouTube).
The feathers of T. rex and other closely related dinosaurs—which were somewhat fluffy, like down feathers—did not have a shape that would have been advantageous for producing aerodynamic lift, which allows both birds and airplanes to stay aloft.
Over the past 20 years, paleontologists have discovered widespread evidence of different kinds of feathers throughout the dinosaur family tree (or phylogeny). One group of theropods, the Dromaeosauridae (which includes the raptors of Jurassic Park fame), were likely entirely feathered and some like Microraptor might have been able to glide or even fly.

Examples of six different kinds of dromaeosaurs. Image by Fred Wierum (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 4.0 International license).

The theropod dinosaur Microraptor gui from the Cretaceous of China. Arrows indicate preserved feathers. Image from Hone et al. (2010) in PLoS ONE 5(2):e9223 (Creative Commons Attribution 2.5 Generic license).
Theropod dinosaurs are the ancestors of today's birds and it has become clear that they inherited their feathers from these dinosaurs. The first feathers in dinosaurs were likely adaptations for thermoregulation (keeping a constant body temperature), not flight.
Reptiles are cold-blooded, meaning that they cannot regulate their own body temperature. They rely on external conditions to warm up. Think of a crocodile basking in the morning sun before it can actively move around.

A cold-blooded Australian saltwater crocodile (Crocodylus porosus) basking in the sunlight. Photograph by Jonathan R. Hendricks.
Dinosaurs that had insulating, heat-trapping feathers would not need to rely as much upon external conditions to regulate their body temperatures. It is very conceivable that the origin of feathers might have provided some dinosaurs with an evolutionary advantage relative to other organisms that lacked them.
After functioning for millions of years as thermoregulatory structures, feathers were later co-opted for flying in dinosaurs. The precise point when they became used for flight marks the transition from non-avian dinosaurs to birds. The fact that feathers start out as a trait that has one function (thermoregulation) and are later modified and co-opted as a trait for another function (flying) helps us understand the manner in which natural selection works. Traits or features are not built wholesale, nor do they necessarily arise to perform the function(s) that they perform today. For this reason the term adaptation is not the preferred term when we refer to structures that shift function over evolutionary history. Instead, these features are called exaptations (a term coined by Elisabeth Vrba and Stephen Jay Gould).
The example of the evolution of feathers demonstrates that the selective regime shaping a trait today may not be the same as the selective regime that shaped that trait initially. This gives us more insight into the way evolution works and suggests that the view of evolution as being solely about the process of adaptation is somewhat simplistic.
In short, having the historical context for understanding traits and their evolution is critical. That’s why a phylogeny is needed in order to show the evolutionary context of a given trait and how it has been modified through time. This makes it possible to determine if the trait is an adaptation or an exaptation. If a trait evolved a long time ago and its function has changed over time, then that trait is an exaptation and not an adaptation.
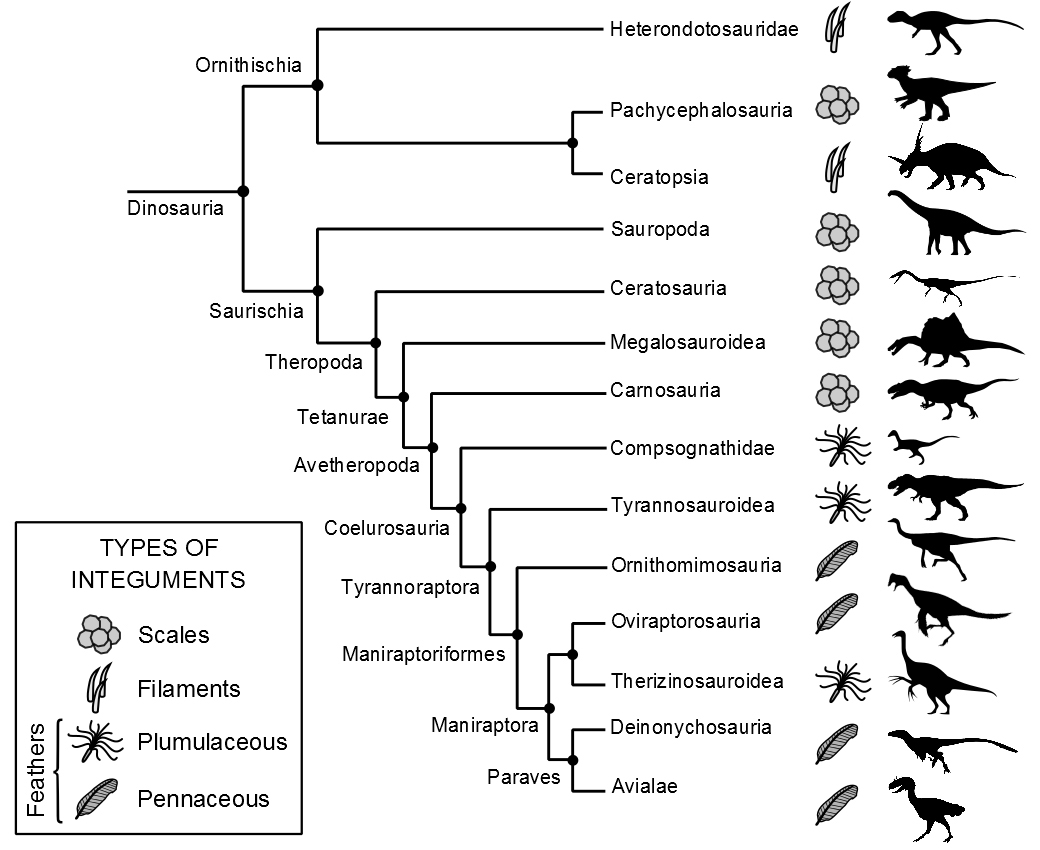
Phylogenetic distribution of different feather types in dinosaurs. Avialae is the group that includes modern birds. Image by Gustavo et al. (2015; pre-print, not yet peer reviewed) (Wikipedia/Wikimedia Commons; Creative Commons Attribution 4.0 International license).
Spandrels
We tend to think of the features of organisms as being functional solutions to particular problems: either adaptations or exaptations. Not all traits, however, are subject to selection.
Some features of organisms arise as a structural byproduct or a side consequence of something else. A great example of this comes from the work of Stephen Jay Gould, who considered the case of the extinct Irish Elk, Megaloceros giganteus.

Two specimens of Irish Elk (Megaloceros giganteus). Left: specimen at the Naturhistorisches Museum Basel, Switzerland. Right: specimen at the American Museum of Natural History, New York City. Photographs by Jonathan R. Hendricks.
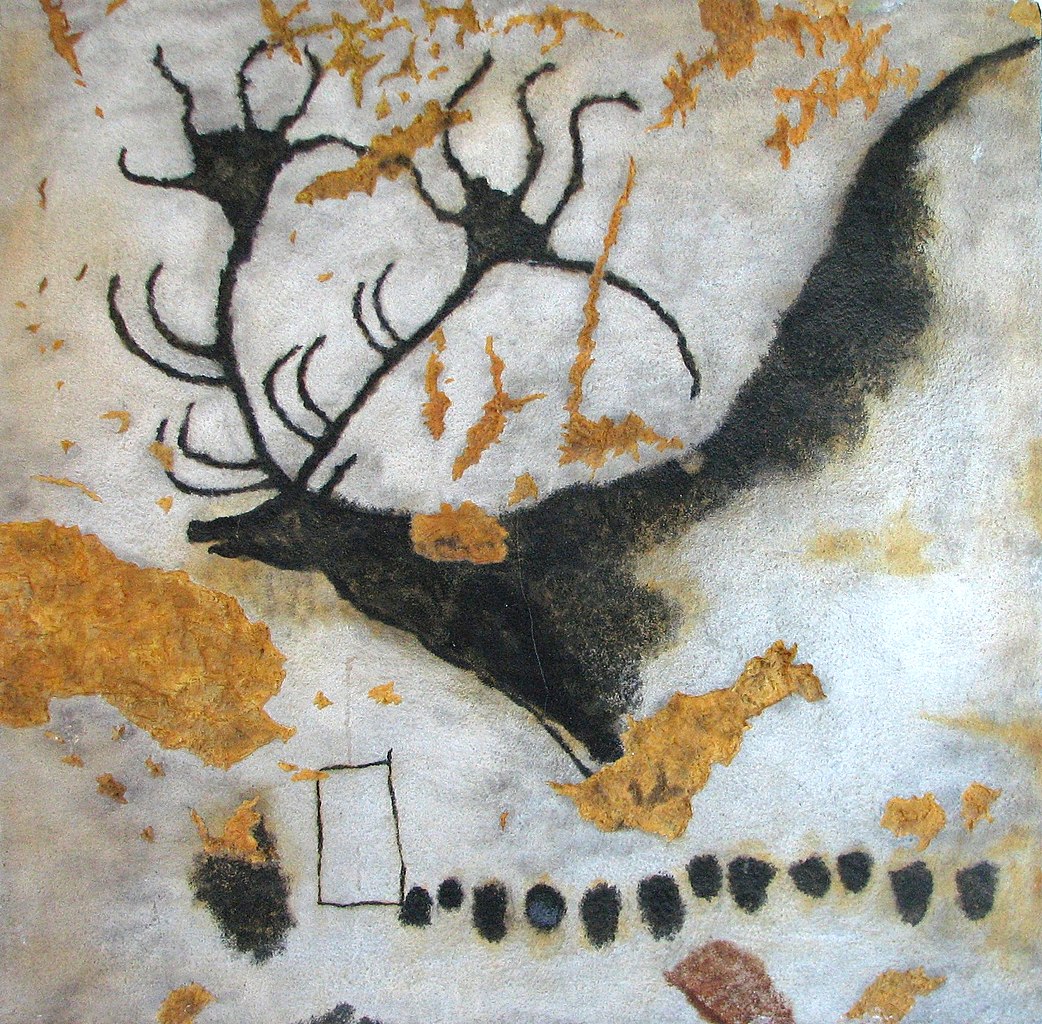
17,000 year-old cave painting of an Irish Elk, from Lascaux, France (Wikipedia/Wikimedia Commons; public domain).
The Irish Elk was not actually an elk, but rather a deer, and its remains are found in many locations around the world, including Ireland. The species went extinct about 8,000 years ago. One of the things that distinguishes the Irish Elk from modern deer is its very large body size and also the truly stupendous size of its rack of antlers.
Evolutionary perspectives that focused on adaptation and natural selection tried to concoct explanations for why the antlers of the Irish Elk were so huge. For example, arguments were made that sexual selection resulted in females preferentially mating with the males with the largest antlers, driving an increase in antler size over time. While it is certainly possible that male Irish Elk fought each other to win mates—and that larger racks helped males to win such fights—we have no evidence from the fossil record to assert anything about such putative pre-historic melees and their consequences for rack size. The “big antlers adaptation hypothesis” was nothing more than an assumption.
The scientific process emphasizes testability and when Gould tested the hypothesis of adaptation in Irish Elk, he found it lacking. Instead, he identified a phenomenon that we think is far more interesting. Gould showed that across all modern deer species, there is a predictable relationship between the body size of the deer and the size of its antlers: as body size increases in deer, the size of their antlers increases much more rapidly, such that large deer have very large antlers (see Fig. 1 in Gould, 1974, reproduced below).
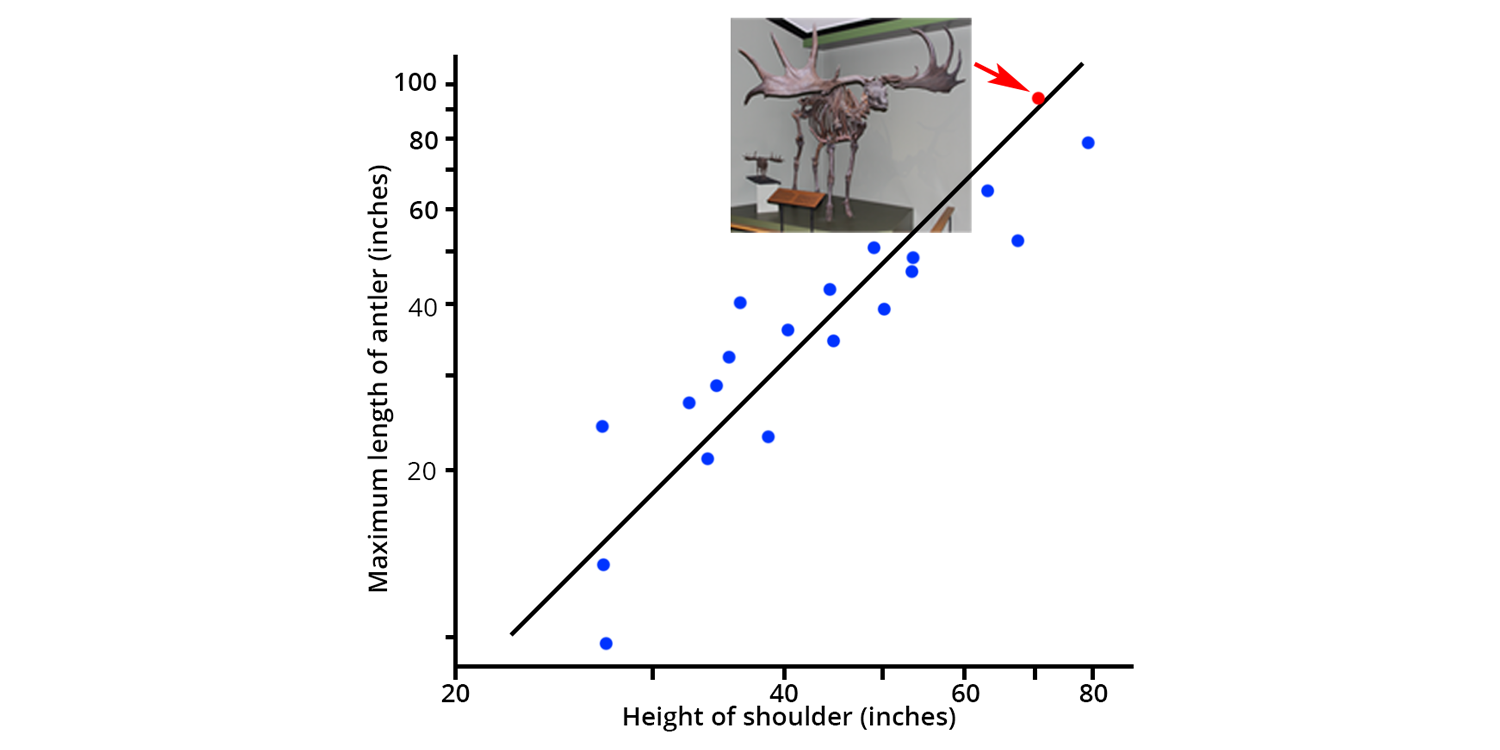
Generalized reproduction of fig. 1 in Gould (1974): "Positive allometry of antlers in cervine deer. Each point represents a single species; data from Ward, 1928. ... Logarithmic scales, in inches" (p. 195). Blue points represent extant deer. The Irish Elk is represented by the red point indicated by the arrow. Image by Jonathan R. Hendricks.
When Gould added the antler and body size data for the Irish Elk, he found that it fit precisely on the regression line that described the relationship between body size and antler size (see figure above). It turns out that Irish Elk were the largest deer that ever lived, and had exactly the right size of antlers given their body size.
There seems to be a predictable relationship in the deer lineage between body size and antler size that has persisted for millions of years. Rather than antler size being a trait honed by natural selection, it seems to be a trait associated with body size. That said, it is certainly possible that there was selection for ever larger body size within the ancestors of the Irish Elk.

Reconstruction of the Irish Elk by famous paleo-artist Charles Knight. Painting is on display at the American Museum of Natural History. Wikipedia/Wikimedia Commons (public domain).
The Irish Elk evolved and lived during the geologically recent Pleistocene Ice Ages, which includes some of the coldest periods that the Earth has experienced in the last 500 million years. It is a well-known fact that larger body size is positively selected in mammals as temperature declines, favoring large size. Thus, large antlers probably evolved as a side consequence of selection for body size in the Irish Elk. In other words, natural selection probably favored larger body size and increasing antler size just "came along for the ride." These large antlers may have subsequently been used for fighting, but they didn’t become large for that purpose.
In 1979, Stephen Jay Gould and Richard Lewontin published a classic paper that argued cogently that we should refer to traits like the antlers of the Irish Elk as “spandrels.”
In architecture, a spandrel is the term for the triangular shape formed when an arch intersects either another arch or a rectangular frame above. They are a simple geometric consequence of how these shapes fit together. Wonderful examples of spandrels can be viewed at Saint Mark's Basilica in Venice, Italy.

Spandrels (indicated by arrows) at Saint Mark's Basilica in Venice, Italy. Photography by "Tango7174" (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 4.0 International license; arrows added to photograph).
At Saint Mark's Basilica, the spandrels are beautifully decorated with paintings, such that they harmoniously fit into the religious allegory that the designers of the cathedral were trying to present. A person unfamiliar with architecture might posit that the spandrels were constructed specifically as attractive triangular canvases to highlight these religious scenes. In reality, the only way for medieval architects to build a cathedral with a large dome was to support that dome via a series of arches. As a consequence, they produced spandrels that were beautifully decorated afterwards. But they did not put the spandrels there on purpose; they had to be there because of the arches and were subsequently utilized for a secondary purpose.
Gould and Lewontin argued persuasively that perhaps many of the features of organisms that scientists call adaptations are in fact spandrels. In other words, perhaps many of the functional structures of organisms are simply structural consequences of other aspects of their forms and were not specifically shaped by natural selection for their current functions.
Intriguingly, even though the classic paper by Gould and Lewontin was published four decades ago (and the ideas therein were further developed in the 1982 paper on exaptation by Gould and Vrba), few, if any, scientists have gone about quantifying the relative proportion of traits that are adaptations versus spandrels. The same can be said for quantification of the proportion of traits that are exaptations, rather than adaptations. These seem like obvious and exciting questions to address, but unfortunately we cannot yet say how frequent these phenomena are in nature. We suspect, however, that they may be commonplace.
Imperfection in nature
Natural selection provides an explanation for why organisms are seemingly so well adapted to the environments in which they live. Consider squirrels: their fur color often provides excellent camouflage against tree bark, their teeth allow them to break open hard nuts with ease, and their tails provide them with the balance that they need to race along telephone wires.

The eastern grey squirrel, Sciurus carolinensis, enjoying a nut in New York City. Photograph by Jonathan R. Hendricks.
Prior to the work of Darwin and Wallace, many naturalists considered such examples of adaptation to be evidence for divine creation (a prime example being Rev. William Paley, who in 1802 wrote a book titled Natural Theology: Or, Evidences of the Existence and Attributes of the Deity). This school of thought (natural theology) survives on today as intelligent design, even though it has not been accepted in any scientific circles since the late 1800s. (A recent attempt to allow the teaching of intelligent design as an alternative to evolution in U.S. public schools was ruled unconstitutional in the 2005 case of Kitzmiller v. Dover Area School District.)
When young, eager naturalists (including Charles Darwin) began looking for examples of divine perfection in nature, they instead found many examples of the opposite: organisms that are good enough to survive, but far from perfect in their design.
Honey bees provide a great example of imperfect design in nature (as recognized by Darwin himself). When they sting some large enemies (such as humans) to defend their hives, a portion of their abdomen is torn from their body with the stinger, causing them to die. If a bee were perfectly created from scratch, wouldn't the design of its stinger allow it to protect its hive over and over again without suffering so?

When a honey bee stings a person, a portion of its abdomen is torn from its body, resulting in its death. Photograph by "Waugsberg" (Wikipedia/Wikimedia Commons; Creative Commons Attribution-Share Alike 3.0 Unported license).
We covered adaptations of the human eye earlier on this page. As it turns out, our own eyes are far from perfect (and don't get us started on how much worse they really are than the much more amazing eyes of the ocean-dwelling mantis shrimp!).
In fact, one of the peculiarities of human vision is that the image that passes through the front of the eye appears upside down on the retina. As shown in the video below, our brains have to translate that information as it comes in from the optic nerve and then reorient everything in the correct position. Luckily, this is done automatically by the brain and requires no thought on our part (thank goodness!). Even so, this does not really seem like a sensible way for our vision to work, but it gets the job done anyway.
"How the Eye Works Animation" by AniMed (YouTube).
Another example of imperfect design in nature is found in the panda bear. Pandas use their thumb to help them strip away the outer layer of bamboo stalks so that they can get at the soft shoot inside, which is their preferred food. Upon close inspection, however, we see that the "thumb" is not an articulated, movable digit at all, but is rather a modified wrist bone.

The panda's "thumb." Left: a panda bear at the Toronto Zoo (Ailuropoda melanoleuca) holding a piece of bamboo by using its modified wrist bone "thumb." Image by Rick Ligthelm (Wikimedia Commons; Creative Commons Attribution 2.0 Generic license). Right: skeleton of a panda bear on display at the Natural History Museum, London. The modified wrist bone is artificially painted blue. Note that the panda bear has five regular digits, just like we do. Image of panda skeleton by Jonathan R. Hendricks.
This is an example of natural selection making do with the materials already available. Certainly if one were to design a panda bear from scratch, it would be given a much more dexterous, articulated thumb digit. While rudimentary, the panda's "thumb" is much more useful than having no thumb at all and must have conferred an important survival advantage after this variation first appeared. This example was made famous by Stephen Jay Gould in his 1980 collection of essays, "The Panda's Thumb."
Whether you consider a bee's stinger, the human eye, or the panda's "thumb," all are good enough to get the job done, but let's not kid ourselves that every structure is perfectly adapted for the task at hand (please do pardon the pun!).
When you’re up in bear country, one of the things that they tell you is you do not need to be an incredibly fast person or faster than a bear to escape one if it decides to attack your party. Instead, you simply have to be faster than the slowest person in your group.
The same is true of natural selection. A trait need only confer a very tiny advantage to its bearer (again, pardon the pun)—relative to the condition in other members of the population that lack the trait—to be positively selected for. Perfection is not necessary and, as Darwin stated in an 1857 letter to Asa Gray, "What a trifling difference must often determine which shall survive, and which perish!"
Indeed, one point that Stephen Jay Gould further made as part of his discussion of the panda’s "thumb" is that these imperfect structures provide some of the best pieces of evidence for evolution because they make it eminently possible for us to trace the historical pattern of descent with modification. They thereby allow us to recognize that evolution, or descent from a common ancestor, happens. Panda’s, for instance—although highly specialized for their vegetarian, bamboo-loving habit, a diet distinct from that of all other bears—are still just bears, albeit awfully cute ones.
"Sneezing Baby Panda" by Wild Candy (YouTube).
References and further reading
Anderson, B.M., and W.D. Allmon. 2018. When domes are spandrels: on septation in turritellids (Cerithioidea) and other gastropods. Paleobiology 44(3): 444-459.
Bowler, P.J. 2009. Evolution: the history of an idea (25th anniversary edition). University of California Press, Berkeley.
Brooks, J.L. 1984. Just Before the origin: Alfred Russell Wallace's theory of evolution. Columbia University Press, New York.
Carroll, S.B. 2009. Remarkable creatures: epic adventures in the search for the origins of species. Mariner Books.
Coghil, L.M., C.D. Hulsey, J. Chaves-Campos, F.J. García de Leon, and S.G. Johnson. 2014. Next generation phylogeography of cave and surface Astyanax mexicanus. Molecular Phylogenetics and Evolution 79: 368-374.
Darwin, C. 1859. On the origin of species by means of natural selection (facsimile of 1st edition). Harvard University Press, Cambridge, Massachusetts.
Darwin, C., and Wallace, A. 1858. On the tendency of species to form varieties; and on the perpetuation of varieties and species by natural means of selection. Journal of the Proceedings of the Linnean Society 3: 45-62.
Desmond, A. 1986. The politics of evolution. University of Chicago Press, Chicago, Illinois.
Eldredge, N. 2005. Darwin: discovering the tree of life. W.W. Norton, New York, New York.
Gould, S.J. 1974. The origin and function of "bizarre" structures: antler size and skull size in the "Irish Elk," Megaloceros giganteus. Evolution 28: 191-220.
Gould, S.J. 1992. The panda's thumb: more reflections in natural history. W.W. Norton & Company, Inc.
Gould, S.J., and R.C. Lewontin. 1979. The spandrels of San Marco and the Panglossian paradigm: a critique of the adaptationist programme. Proceedings of the Royal Society of London, B 205: 581-598.
Gould, S.J., and E.S. Vrba. 1982. Exaptation—a missing term in the science of form. Paleobiology 8(1): 4-15.
Hone, D.W.E., H. Tischlinger, X. Xu, and F. Zhang. 2010. The extent of the preserved feathers on the four-winged dinosaur Microraptor gui under ultraviolet light. PLoS ONE 5(2): e9223.
Jacob, F. 1977. Evolution and tinkering. Science 196(4295): 1161-1166.
Lieberman, B.S. 2000. Paleobiogeography: using fossils to study global change, plate tectonics, and evolution. Kluwer Academic/Plenum Publishing, New York, New York.
Lieberman, B.S., and R.L. Kaesler. 2007. Prehistoric life: evolution and the fossil record. John Wiley & Sons, New York, New York.
Marshall, E. 2018. 1st July 2018: 160th anniversary of the presentation of "On the tendency of Species to form Varieties." The Linnean Society of London News (blog).
Mayr, E. 1982. The growth of biological thought. Harvard University Press, Cambridge, Massachusetts.
Osborn, H.F. 1903. From the Greeks to Darwin: an outline of the development of the evolution idea. MacMillan, London.
Parker, H.G., D.L. Dreger, M. Rimbault, B.W. Davis, A.B. Mullen, G. Carpintero-Ramirez, and E.A. Ostrander. 2017. Cell Reports 19(4): P697-708.
Prothero, D.R. 2008. Evolution: what the fossils say and why it matters. Columbia University Press, New York, New York.
Prothero, D.R. 2015. The story of life in 25 fossils. Columbia University Press, New York, New York.
Shearin, A.L. and E.A. Ostrander. 2010. Canine morphology: hunting for genes and tracking mutations. PLoS Biology 8(3): e1000310.
van Oosterzee, P. 1997. Where worlds collide: the Wallace Line. Cornell University Press, Ithaca, New York.
Wallace, A.R. 1855. On the law which has regulated the introduction of new species. The Annals and Magazine of Natural History, Ser. 2, 16:184-196.
Wallace, A.R. 1869. The Malay Archipelago (facsimile edition). Dover Press, New York, New York.
Winsor, M. 1991. Reading the shape of nature: comparative zoology at the Agassiz Museum. University of Chicago Press, Chicago, Illinois.
Content usage
Usage of text and images created for DEAL: Text on this page was written by Bruce S. Lieberman and Jonathan R. Hendricks. Original written content created by Bruce S. Lieberman and Jonathan R. Hendricks for the Digital Encyclopedia of Ancient Life that appears on this page is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. Original images created by Jonathan R. Hendricks are also licensed under Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Content sourced from other websites: Attribution, source webpage, and licensing information or terms of use are indicated for images sourced from other websites in the figure caption below the relevant image. See original sources for further details. Attribution and source webpage are indicated for embedded videos. See original sources for terms of use. Reproduction of an image or video on this page does not imply endorsement by the author, creator, source website, publisher, and/or copyright holder.



